Myocardial Ischemia & infarction: Cellular changes, ECG and symptoms
Myocardial ischemia: cellular changes, ECG signs and development of infarction
Like all other cells in the human body, cardiac myocytes use ATP (adenosine triphosphate) as the primary energy source. ATP is produced by metabolizing carbohydrates (glucose), fats or proteins, whichever is available. ATP fuels all cellular functions, such as contraction and relaxation. Increasing cardiac workload by increasing heart rate and contractility (i.e contractile force), results in increased metabolism. Cardiac cells are highly capable of matching the supply and demand of ATP; increased cardiac workload results in increased ATP production and vice versa. Importantly, cardiac myocytes require aerobic metabolism (oxygen) to produce sufficient quantities of ATP. This implies that the heart requires continuous blood flow to maintain cellular function.
Myocardial ischemia occurs when there is insufficient oxygen available. Insufficient oxygen supply results in diminished ATP production and subsequently disruption of cellular metabolism. In case of ischemia, cardiac myocytes protect themselves by switching to anaerobic metabolism. This is possible because cardiac myocytes store glycogen (a storage form of glucose) which can be used to produce ATP in absence of oxygen. Unfortunately, glycogen yields only small amounts of ATP and glycogen supplies are limited. In order to reduce oxygen demands, cardiac myocytes stop contracting under anaerobic conditions (i.e during ischemia). These two measures – i.e switching to anaerobic metabolism and discontinuing the contractions – enables the myocardium to endure 20 to 30 minutes of severe ischemia. If myocardial perfusion (blood flow) in the ischemic zone is not restored before that time limit, the cell will die.
Myocardial ischemia in clinical practice: coronary artery disease
In clinical practice, myocardial ischemia occurs in numerous situations. In stable angina pectoris, there are atherosclerotic plaques which limit coronary blood flow and cause symptoms during increased myocardial workload (exercise). The more severe the stenosis, the more pronounced the symptoms. Stable angina pectoris is diagnosed by means of exercise stress testing (exercise ECG). The purpose of the stress test is to increase the myocardial workload (and thus the oxygen demand) in order to provoke ischemia in myocardium supplied by atherosclerotic arteries. This engenders the typical ECG changes as well. In acute coronary syndromes, however, symptoms and ECG changes are manifest at rest because of the severe reduction of coronary flow caused by acute atherothrombosis.
Table 1: Myocardial and ECG reaction in various settings with ischemia or increased workload
| STATUS OF CORONARY ARTERY | SETTING | EFFECT | ECG REACTION |
| Normal coronary artery (no atherosclerosis) | At rest | Oxygen supply is adequate and ischemia cannot occur. | No changes on resting ECG. |
| Increased myocardial workload (exercise) | Oxygen supply increases in parallel with increased oxygen consumption during exercise. Thus, supply and demand are balanced and no ischemia occurs. (Some individuals display a benign form of ischemia located in the subendocardium during exercise; this manifests with ST-segment depressions with upsloping ST-segment during exercise but the person is asymptomatic). | No changes on resting ECG. Benign ST-segment depression has an upsloping ST-segment during exercise stress testing. | |
| Stable coronary artery disease (atherosclerotic plaque causing 70% stenosis or more) | At rest | No symptoms. | No changes on resting ECG. |
| Increased myocardial workload (exercise) | Oxygen demand increases with workload but the stenosis (atherosclerotic plaque) limits the needed increase in blood flow, which results in myocardial ischemia. The ischemia manifests as chest pain. This type of ischemia (induced by increased workload) is located in the subendocardial muscle layer. It is reversible and will resolve when the exercise is stopped. Only on rare occasions (if the ischemia is severe and prolonged) does this result in troponin leakage (i.e cell death). | Exercise stress testing is used to provoke the same ischemic process and this may reveal ST segment depressions, diminished T-wave amplitude and, on rare occasions, ST-segment elevations. | |
| Stable but severe coronary artery disease (atherosclerotic plaque causing 90% stenosis or more) | At rest | Oxygen supply increases in parallel with increased oxygen consumption during exercise. Thus, supply and demand is balanced and no ischemia occurs. (Some individuals display a benign form of ischemia located in the subendocardium during exercise; this manifests with ST-segment depressions with upsloping ST-segment during exercise but the person is asymptomatic). | Resting ECG may show ST-deviation and/or T-wave changes. |
| Plaque rupture/erosion with ensuing acute coronary syndrome | Any time | Transmural ischemia causes ST-segment elevation. Subendocardial ischemia causes ST-segment depressions. Symptoms are pronounced (particularly in transmural ischemia) and are not alleviated by resting or administering nitroglycerin. | Resting ECG shows ST-deviation, T-wave changes and occasionally QRS changes |
| Any status of coronary artery | Coronary artery vasospasm | Severe atherosclerosis may cause ischemia already at rest. This is a serious condition that lies between stable atherosclerosis and acute coronary syndrome. | Resting ECG usually shows ST-segment elevations which indicates that the ischemia is transmural |
Time is muscle: 30 minutes from myocardial ischemia to infarction
The duration of ischemia is crucial when an occlusion has occurred. Myocardium supplied by the occluded artery immediately becomes ischemic and ceases to contract. As mentioned above, the cells revert to anaerobic metabolism to maintain viability. This enables the cell to endure 20–30 minutes of ischemia. If coronary flow is restored within that period, all ischemic myocardium will recover (after a brief period of contractile dysfunction [called stunned myocardium]). If coronary flow is not restored, infarction will commence and the necrosis will spread like a wavefront in water, starting in the most ischemic area, which is the subendocardium. From there the infarction will spread towards the epicardium. Refer to Figure 2.
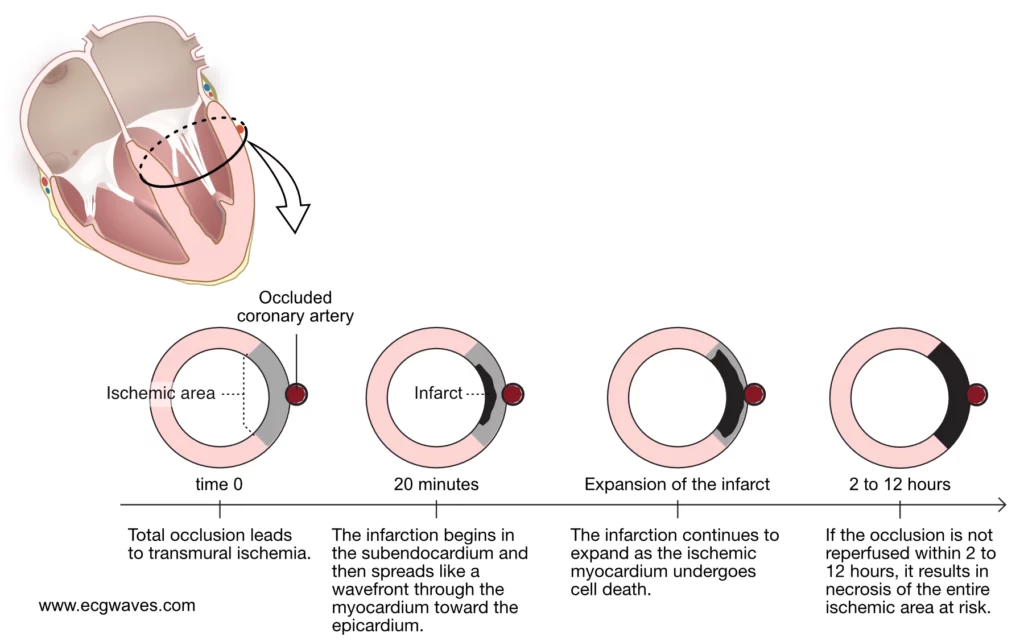
The time it takes for all ischemic myocardium to become infarcted is of great interest. In the literature it is traditionally suggested that the infarction is completed within 4 to 6 hours but this is questioned by newer studies which suggest longer durations. Recent studies actually suggest that the infarction may be completed anywhere between 2 to 12 hours after onset of symptoms. This wide time range is due to factors that modify the natural course. One such factor is the presence of collateral coronary circulation.
Total occlusions (which result in acute STEMI) are generally persistent until virtually all ischemic myocardium is infarcted (unless reperfusion therapy is successful). One-third of all total occlusions are recanalized spontaneously within 12–24 hours (the coronary arteries have a thrombolytic system that handles the thrombus, albeit too late). It should be noted, however, that although the vast majority of the affected myocardium will be necrotic by the time the artery is recanalized, restoration of blood flow may improve contractile function and prognosis. This is because there may be hibernating myocardium in or near the affected area. Hibernating myocardium is severely ischemic but viable and may recover fully if blood flow is restored. Moreover, restoration of blood flow improves healing of the infarct area (which yields a stronger scar) and slows ventricular remodeling (which leads to heart failure).
Figure 3 illustrates how the infarction spreads from the subendocardium towards the epicardium. The reason why the infarction starts in the subendocardium is simply because it has the poorest prerequisites in case of ischemia. The subendocardium is located too far away from the ventricular cavity in order to enjoy oxygen from the cavity. Moreover, it receives blood which has already been extracted of much of its oxygen as it has passed through the bulk of the ventricular wall (coronary blood flow is directed from the epicardium to the endocardium (Figure 3).


