Introduction
Ventricular tachycardia (VT) represents a complex and potentially lethal spectrum of cardiac arrhythmias that remains a formidable challenge in contemporary cardiovascular medicine. As a primary driver of sudden cardiac death (SCD) and a major cause of morbidity in patients with structural heart disease, VT necessitates a nuanced, multidisciplinary approach to diagnosis and management. Over the past decade, the landscape of VT management has undergone a profound paradigm shift. Driven by advanced electroanatomical mapping, high-resolution cardiac magnetic resonance (CMR) imaging, and landmark clinical trials, the field has transitioned from a predominantly pharmacological strategy to one that heavily integrates early catheter ablation and sophisticated device therapy [1][2].
This exhaustive review provides a comprehensive synthesis of the current state of VT management for the practicing cardiologist. It meticulously details the classification, pathophysiological subtypes, and clinical presentation of VT, with a deep dive into the underlying etiologies ranging from benign idiopathic variants to complex scar-related and genetic substrates. Furthermore, it critically examines the diagnostic algorithms for wide-complex tachycardia (WCT), the acute management of both stable and unstable presentations, the critical entity of electrical storm, and the long-term management strategies informed by recent sub-group analyses, registry data, and ongoing clinical trials.
1. Ventricular Tachycardia: Classification, Underlying Etiologies, and Clinical Presentation
Classification of Ventricular Tachycardia
The classification of VT is fundamentally based on its duration, morphology, and hemodynamic consequences, which collectively dictate the urgency and modality of intervention [3].
- By Duration:
- Non-sustained VT (NSVT): Defined as three or more consecutive ventricular beats at a rate exceeding 100 beats per minute (bpm), which terminates spontaneously within 30 seconds [1]. While often asymptomatic, NSVT serves as a critical marker of arrhythmogenic risk, particularly in the setting of ischemic heart disease or reduced left ventricular ejection fraction (LVEF).
- Sustained VT: Defined as VT lasting longer than 30 seconds or requiring termination earlier due to hemodynamic collapse [4]. Sustained VT is a medical emergency that universally warrants acute intervention and long-term secondary prevention strategies.
- By Morphology:
- Monomorphic VT (MMVT): Characterized by a uniform QRS morphology from beat to beat in each electrocardiographic lead. MMVT strongly indicates a stable, fixed arrhythmogenic substrate, most commonly a macro-reentrant circuit anchored around myocardial scar tissue [5].
- Polymorphic VT (PMVT): Characterized by a continuously changing QRS morphology and axis, indicating a dynamic sequence of ventricular activation. PMVT is frequently associated with acute myocardial ischemia, electrolyte derangements, or underlying channelopathies (e.g., Torsades de Pointes in Long QT Syndrome) [1].
Underlying Etiologies
The etiology of VT is broadly dichotomized into structural heart disease (SHD) and structurally normal hearts, a division that profoundly influences prognosis and therapeutic decision-making.
Structural Heart Disease (SHD): Accounting for approximately 90% of all VT presentations, SHD provides the macroscopic architectural abnormalities necessary for arrhythmogenesis. Ischemic heart disease (IHD), particularly prior myocardial infarction (MI), is the most prevalent etiology. The necrotic myocardium is replaced by dense collagenous scar, interspersed with surviving bundles of myocytes that form the slow-conducting isthmuses critical for re-entry [6]. Non-ischemic dilated cardiomyopathy (NICM), hypertrophic cardiomyopathy (HCM), and arrhythmogenic right ventricular cardiomyopathy (ARVC) also fall under this umbrella, each presenting unique fibrotic patterns and arrhythmogenic mechanisms [2].
Structurally Normal Hearts: Comprising the remaining 10% of cases, these VTs occur in the absence of macroscopic myocardial disease. They include idiopathic VTs, which typically originate from specific anatomical regions such as the ventricular outflow tracts or the Purkinje system, and primary electrical disorders (channelopathies) such as Long QT Syndrome (LQTS), Brugada Syndrome, and Catecholaminergic Polymorphic VT (CPVT) [7][8].
Clinical Presentation
The clinical presentation of VT spans a remarkably wide spectrum, dictated primarily by the ventricular rate, the duration of the arrhythmia, the underlying LVEF, and the integrity of peripheral compensatory mechanisms [1]. Patients with preserved LVEF and slower VT rates (e.g., 120-150 bpm) may remain entirely asymptomatic or report mild palpitations and lightheadedness. Conversely, rapid VT in the setting of severe left ventricular dysfunction frequently precipitates presyncope, syncope, ischemic chest pain, or acute heart failure exacerbation due to the sudden loss of atrioventricular synchrony and precipitous drop in cardiac output [4].
At the most severe end of the spectrum, VT presents as profound cardiogenic shock, hemodynamic collapse, and sudden cardiac death (SCD). In these instances, the rapid ventricular rate precludes adequate diastolic filling, leading to an immediate cessation of effective systemic perfusion [9].
2. Subtypes in Relation to Emergence, Persistence, Morphology, and Etiology
Understanding the specific subtypes of VT is paramount, as their emergence, morphological characteristics, and persistence are intimately linked to their underlying cellular mechanisms and etiologies.
Benign Variants: Idiopathic Ventricular Tachycardias
Idiopathic VTs occur in patients without structural heart disease and generally carry a benign prognosis regarding sudden cardiac death, though they can cause significant symptom burden and, rarely, tachycardia-induced cardiomyopathy [7].
- Right Ventricular Outflow Tract (RVOT) VT:
- Emergence and Etiology: RVOT VT is the most common idiopathic VT. Its emergence is typically triggered by states of high sympathetic tone, such as exercise, emotional stress, or caffeine consumption. The underlying cellular mechanism is cyclic AMP (cAMP)-mediated intracellular calcium overload, leading to delayed afterdepolarizations (DADs) and triggered activity [7].
- Morphology: Because the activation originates in the anterior, superior right ventricle and spreads inferiorly and to the left, the surface ECG demonstrates a classic Left Bundle Branch Block (LBBB) pattern with a strong inferior axis (tall, monophasic R waves in leads II, III, and aVF) [10].
- Persistence: It frequently presents as repetitive monomorphic NSVT or sustained VT. Crucially, due to its cAMP-dependent mechanism, it is highly sensitive to termination by intravenous adenosine or beta-blockers [11].
- Left Ventricular Outflow Tract (LVOT) VT:
- Emergence and Etiology: Shares a similar triggered-activity mechanism and sympathetic emergence profile with RVOT VT. Anatomically, it originates from structures such as the aortic sinuses of Valsalva, aortomitral continuity, or epicardial LV summit.
- Morphology: Can present with either an LBBB or RBBB pattern depending on the exact origin. It is distinguished from RVOT VT by an earlier precordial transition (R wave becomes larger than the S wave by lead V1 or V2), reflecting its more posterior and leftward anatomical origin [12].
- Fascicular VT (Belhassen Tachycardia):
- Emergence and Etiology: Typically emerges in young, healthy males (15-40 years) at rest or during mild exercise. Unlike outflow tract VTs, the mechanism is localized micro-reentry within the specialized Purkinje conduction system, utilizing a slow-conducting calcium-dependent tissue as the antegrade limb [13].
- Morphology: Because it exits the Purkinje system into the left ventricle, it presents with a Right Bundle Branch Block (RBBB) pattern. Approximately 90% originate from the left posterior fascicle, resulting in left axis deviation. A minority originate from the left anterior fascicle (right axis deviation) [14].
- Persistence: Often sustained and highly symptomatic. It is uniquely sensitive to intravenous verapamil, which blocks the calcium-dependent slow pathway of the re-entrant circuit [15].
Ischemia and Scar-Related VT
Ischemic VT is the quintessential scar-related arrhythmia. Its emergence typically occurs months to years following a myocardial infarction, as the necrotic tissue undergoes fibrotic remodeling [6].
- Etiology and Mechanism: The mechanism is fixed anatomical macro-reentry. The dense, unexcitable collagenous scar forms the central obstacle. Surviving bundles of myocytes at the scar border zone, often separated by strands of fibrosis, exhibit depressed excitability and anisotropic (directionally dependent) slow conduction. This creates the perfect milieu for a re-entrant circuit: an area of unidirectional block, a slow-conducting protected isthmus, and an exit site into healthy myocardium [5].
- Morphology: Typically monomorphic. The specific QRS morphology dictates the exit site of the circuit from the scar. For example, an anterior wall scar with an exit site in the anterior LV will produce an RBBB pattern with an inferior axis.
- Persistence: Ischemic VT is highly persistent and recurrent. Once the anatomical substrate is formed, it remains a lifelong risk. It is notoriously refractory to antiarrhythmic drugs and frequently requires implantable cardioverter-defibrillator (ICD) therapy and catheter ablation [16].
Cardiomyopathies and Heart Failure
Non-ischemic structural diseases present diverse arrhythmogenic substrates.
- Dilated Cardiomyopathy (DCM): Pathological remodeling leads to patchy, mid-wall or epicardial interstitial fibrosis, creating substrates for scar-related re-entry. Additionally, DCM patients are uniquely prone to Bundle Branch Re-entrant VT (BBR-VT), a macro-reentrant circuit utilizing the right and left bundle branches. BBR-VT typically presents with a rapid, typical LBBB morphology and is highly amenable to curative ablation of the right bundle branch [17].
- Hypertrophic Cardiomyopathy (HCM): Characterized by profound myocyte hypertrophy, severe myofibrillar disarray, and microvascular ischemia leading to replacement fibrosis. This chaotic architecture creates a highly arrhythmogenic substrate. HCM is a leading cause of SCD in young athletes, often presenting with the sudden emergence of PMVT or ventricular fibrillation (VF) during exertion [18].
- Arrhythmogenic Right Ventricular Cardiomyopathy (ARVC): A genetic desmosomal disorder characterized by progressive fibrofatty replacement of the right ventricular myocardium. Morphology: VT typically exhibits an LBBB pattern. The resting ECG often reveals Epsilon waves (small deflections at the end of the QRS complex reflecting delayed RV activation) and T-wave inversions in leads V1-V3 [19][20].
- Heart Failure with Reduced Ejection Fraction (HFrEF): Regardless of the initial insult, the progression to HFrEF involves profound structural remodeling, mechanical wall stretch, and chronic neurohormonal activation (sympathetic nervous system and renin-angiotensin-aldosterone system). These factors alter ion channel expression (e.g., downregulation of repolarizing potassium currents), prolonging action potential duration and lowering the threshold for VT emergence. VT in HFrEF is associated with high persistence and significantly increased mortality [21].
Genetic and Electrical Disorders (Channelopathies)
These disorders involve mutations in genes encoding cardiac ion channels or associated regulatory proteins, leading to primary electrical instability without macroscopic structural disease [8].
- Long QT Syndrome (LQTS): Characterized by delayed myocardial repolarization. Emergence: Triggers are genotype-specific: sudden adrenergic surges or swimming in LQT1 (KCNQ1 mutation), sudden auditory stimuli in LQT2 (KCNH2 mutation), or during sleep/bradycardia in LQT3 (SCN5A mutation). Morphology: The hallmark is Torsades de Pointes (TdP), a PMVT characterized by a continuously twisting QRS axis around the isoelectric line, driven by early afterdepolarizations (EADs) [22].
- Brugada Syndrome: An autosomal dominant disorder linked to sodium channel (SCN5A) loss-of-function mutations. Emergence: VT/VF typically emerges during sleep, rest, or febrile states. Morphology: Presents as PMVT or VF. The diagnostic resting ECG shows a coved-type ST-segment elevation ≥2 mm followed by a negative T wave in the right precordial leads (V1-V2) [23].
- Catecholaminergic Polymorphic VT (CPVT): Caused by mutations in the ryanodine receptor (RYR2) or calsequestrin (CASQ2), leading to spontaneous diastolic calcium leak from the sarcoplasmic reticulum. Emergence: Strictly triggered by physical exercise or acute emotional stress. Morphology: Classically presents as bidirectional VT (alternating QRS axis beat-to-beat) or rapid PMVT [24].
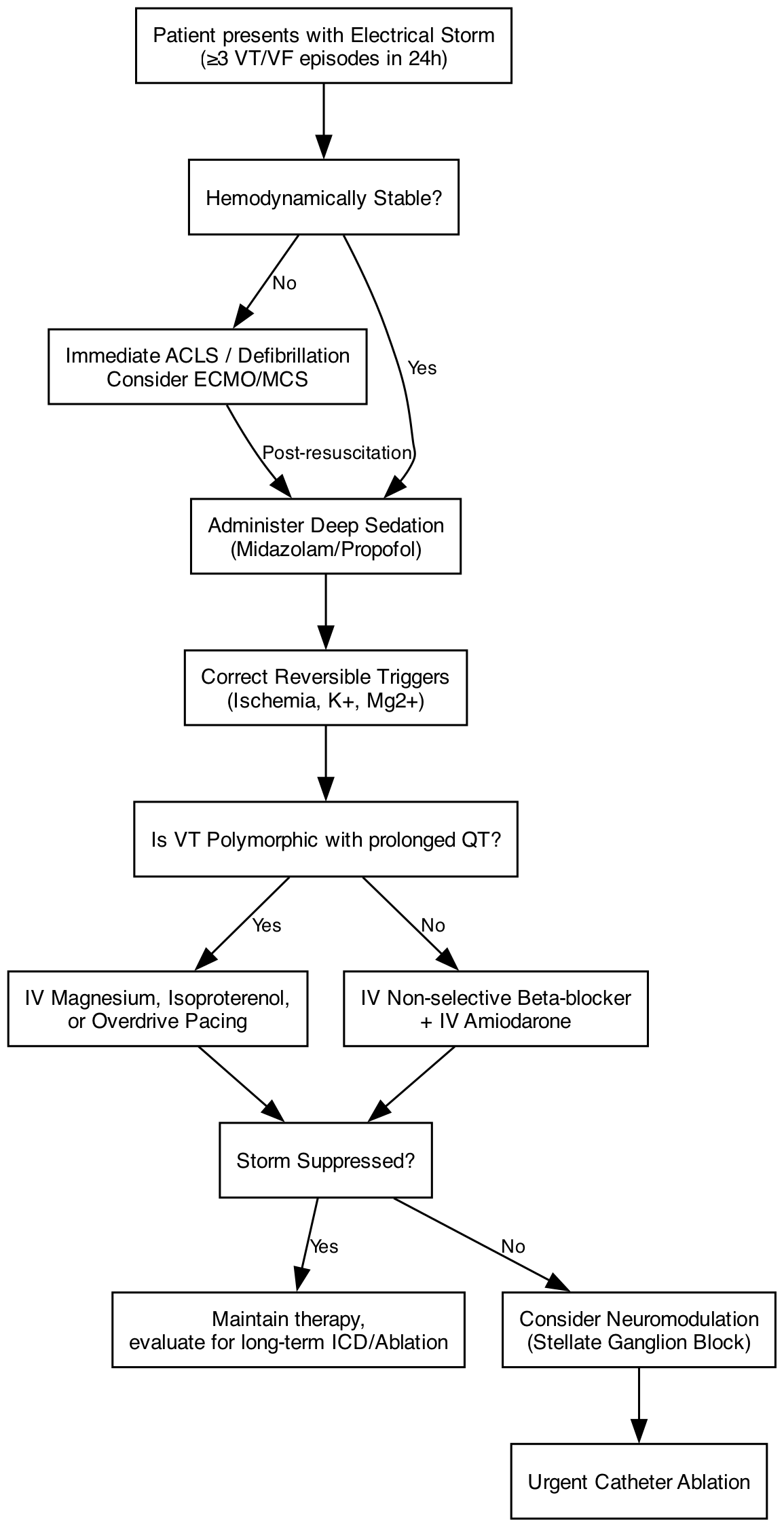
3. Definition and Management of Electrical Storm
Definition and Clinical Significance
Electrical storm (ES), also termed VT storm, is a life-threatening cardiac emergency defined by the occurrence of three or more distinct episodes of sustained VT, VF, or appropriate ICD shocks within a 24-hour period [1][25]. The episodes must be separated by at least 5 minutes to be considered distinct events. ES represents a state of extreme, self-perpetuating cardiac electrical instability. It is associated with profound psychological trauma for the patient, rapid hemodynamic deterioration, and a significantly elevated short-term and long-term mortality rate [26].
Pathophysiology
The emergence of an electrical storm is typically driven by a classic pathophysiological triad [27]:
- Susceptible Substrate: The presence of an underlying arrhythmogenic focus, most commonly a dense ischemic scar with surviving border-zone myocardium.
- Reversible Triggers: Acute physiological derangements that destabilize the substrate, such as acute myocardial ischemia, severe electrolyte imbalances (hypokalemia, hypomagnesemia), acute heart failure decompensation, or drug toxicity.
- Autonomic Dysfunction: A massive, uncontrolled surge in sympathetic nervous system tone. The initial VT episode or ICD shock causes pain and anxiety, triggering a catecholamine release. This sympathetic surge further shortens the ventricular refractory period, enhances conduction velocity heterogeneity, and triggers subsequent VT episodes, creating a lethal positive feedback loop.
Management Strategies
The management of ES requires a rapid, multipronged approach aimed at breaking the sympathetic cascade, suppressing the arrhythmia, and stabilizing hemodynamics [1][25].
- Sedation and Anesthesia: Mild to deep sedation (using agents like midazolam, propofol, or dexmedetomidine) is a critical first-line intervention. In refractory cases, general anesthesia and intubation are required to completely abolish psychological distress and blunt the central sympathetic drive [1].
- Pharmacotherapy:
- Beta-blockers: Non-selective beta-blockers, particularly intravenous propranolol or oral nadolol, are superior to selective agents (like metoprolol) because they block both beta-1 and beta-2 receptors, providing more comprehensive sympathetic blockade [28].
- Antiarrhythmics: Intravenous amiodarone is the cornerstone antiarrhythmic for ES in patients with structural heart disease. If amiodarone fails, intravenous lidocaine can be added, as it specifically targets ischemic, partially depolarized tissue [29]. Crucial Exception: If the storm is driven by PMVT/Torsades de Pointes in the setting of prolonged QT, amiodarone is contraindicated; management requires intravenous magnesium sulfate, overdrive pacing, or isoproterenol [1].
- Neuromodulation: For ES refractory to deep sedation and maximal pharmacotherapy, targeted autonomic modulation is highly effective. Techniques include percutaneous stellate ganglion block (typically left-sided, using bupivacaine) or thoracic epidural anesthesia, which directly interrupt the sympathetic efferent pathways to the heart [30].
- Catheter Ablation: Urgent radiofrequency catheter ablation is now a Class I recommendation for ES refractory to medical therapy. Advanced electroanatomical mapping allows for the identification and homogenization of the arrhythmogenic substrate. Ablation during ES has been shown to significantly improve acute survival and reduce long-term recurrences [31].
- Mechanical Circulatory Support (MCS): In patients presenting with or progressing to severe cardiogenic shock due to incessant VT, temporary MCS (such as venoarterial ECMO or Impella) is required to maintain end-organ perfusion, unload the ventricle, and serve as a bridge to definitive catheter ablation or cardiac transplantation [25].
4. Key ECG Features Supporting VT and Differential Diagnostics of Wide-Complex Tachycardia
The Challenge of Wide-Complex Tachycardia (WCT)
Wide-complex tachycardia is defined by a QRS duration ≥ 120 milliseconds and a heart rate > 100 bpm. The differential diagnosis of WCT is a classic conundrum in emergency cardiology. Statistically, 80% to 90% of all WCTs in adults are VT. The remaining cases comprise [32][33]:
- SVT with Aberrancy: Supraventricular tachycardia conducted to the ventricles with a pre-existing or rate-related (phase 3 block) bundle branch block.
- SVT with Pre-excitation: Antidromic atrioventricular reentrant tachycardia (AVRT) conducting antegrade down an accessory pathway (e.g., Wolff-Parkinson-White syndrome), resulting in a fully pre-excited, wide QRS.
- Pacemaker-Mediated Tachycardia: Or other device-related tachycardias.
- Toxic/Metabolic WCT: Severe hyperkalemia or sodium-channel blocker toxicity (e.g., tricyclic antidepressants) causing profound QRS widening.
Clinical Axiom: In the acute setting, any WCT must be presumed to be VT and treated as such until proven otherwise. Misdiagnosing VT as SVT and administering atrioventricular nodal blocking agents (like verapamil) can precipitate catastrophic hemodynamic collapse [1].
Key ECG Features Supporting VT
Careful analysis of the 12-lead ECG can reliably differentiate VT from SVT with aberrancy [1][32].
- Atrioventricular (AV) Dissociation: The absolute hallmark of VT. It manifests as independent P waves marching through the QRS complexes at a rate slower than the ventricular rate. While highly specific (approaching 100%), its sensitivity is low, as P waves are discernible in only about 30% of WCT surface ECGs [34].
- Fusion and Capture Beats: Direct consequences of AV dissociation. A capture beat occurs when a sinus impulse fortuitously conducts through the AV node and completely depolarizes the ventricles, resulting in a narrow QRS complex amidst the WCT. A fusion beat occurs when a sinus impulse and the VT impulse simultaneously depolarize the ventricles, creating a hybrid QRS morphology. Both definitively prove a ventricular origin of the tachycardia.
- Extreme Axis Deviation: A “northwest axis” (between -90° and -180°, with negative QRS complexes in leads I and aVF) strongly suggests VT, as the activation is proceeding from the apex toward the base of the heart, opposite to normal conduction.
- Precordial Concordance: Occurs when all QRS complexes from V1 to V6 are entirely positive (R waves) or entirely negative (QS complexes). Negative concordance is virtually diagnostic of VT (often apical origin). Positive concordance is highly suggestive of VT (basal origin) but can rarely be seen in antidromic AVRT [1].
- QRS Duration: A QRS duration > 140 ms in the presence of an RBBB morphology, or > 160 ms in the presence of an LBBB morphology, strongly favors VT over SVT with aberrancy [32].
Diagnostic Algorithms
Several stepwise algorithms have been developed to systematize ECG interpretation in WCT.
- The Brugada Algorithm (1991): A classic four-step approach [34].
- Absence of an RS complex in all precordial leads (V1-V6)? If yes → VT.
- If RS is present, is the longest RS interval (onset of R to nadir of S) > 100 ms in any precordial lead? If yes → VT.
- Is AV dissociation present? If yes → VT.
- Are morphological criteria for VT present in leads V1-V2 and V6? If yes → VT. If all four steps are negative, the diagnosis is SVT with aberrancy.
- The Vereckei aVR Algorithm (2008): Focuses solely on lead aVR, simplifying analysis [35].
- Is there an initial R wave in aVR? If yes → VT.
- Is there an initial r or q wave > 40 ms? If yes → VT.
- Is there notching on the initial downstroke of a predominantly negative QRS? If yes → VT.
- Is the Vi/Vt ratio (voltage during the initial 40 ms divided by voltage during the terminal 40 ms) < 1? If yes → VT.
- The Basel Algorithm (2022): A modern, simplified algorithm designed for rapid use by non-electrophysiologists in the emergency department. It requires at least two of the following three criteria to diagnose VT [36][37]:
- Clinical high risk (history of prior MI, heart failure, or presence of an ICD)?
- Lead II time to first peak > 40 ms?
- Lead aVR time to first peak > 40 ms?

5. Management of Hemodynamically Stable VT
Initial Approach and Paradigm Shift
A patient presenting with sustained VT who maintains adequate blood pressure, normal mentation, and lacks signs of severe end-organ hypoperfusion is considered hemodynamically stable. The initial approach involves continuous telemetry, acquisition of a 12-lead ECG, securing intravenous access, and the immediate application of defibrillator pads [1].
Historically, chemical cardioversion was the preferred first-line strategy for stable VT. However, the 2022 European Society of Cardiology (ESC) Guidelines introduced a significant paradigm shift: continuous synchronized electrical cardioversion is now elevated to a first-line therapy (Class I) for stable sustained monomorphic VT, provided the patient can be safely sedated [1]. This shift acknowledges that electrical cardioversion has a near 100% efficacy rate, avoids the proarrhythmic and hypotensive risks of antiarrhythmic drugs, and rapidly restores sinus rhythm, preventing unexpected hemodynamic deterioration.
Chemical Cardioversion: The PROCAMIO Trial
If chemical cardioversion is chosen, the selection of the antiarrhythmic agent is critical. For decades, intravenous amiodarone was the default choice. This practice was fundamentally challenged by the landmark PROCAMIO trial (2017) [38].
PROCAMIO was a randomized, open-label trial comparing intravenous procainamide (10 mg/kg over 20 minutes) to intravenous amiodarone (5 mg/kg over 20 minutes) for the acute treatment of tolerated WCT. The results were definitive:
- Efficacy: Procainamide achieved a significantly higher termination rate of the tachycardia compared to amiodarone (67% vs. 38%, p = 0.026).
- Safety: Procainamide had a significantly lower incidence of major adverse cardiac events (9% vs. 41%, p = 0.001). The primary driver of adverse events in the amiodarone arm was profound hypotension requiring immediate electrical cardioversion or vasopressor support.
Based on these data, intravenous procainamide is now the preferred pharmacological agent for stable monomorphic VT of unknown etiology or known structural heart disease [1][39]. Amiodarone remains an alternative but is associated with delayed onset of action and higher rates of hypotension.
Management of Specific Idiopathic VTs
If the 12-lead ECG and clinical history strongly suggest a specific idiopathic VT, targeted pharmacotherapy is indicated [1]:
- Fascicular VT: Intravenous verapamil is the drug of choice, given the calcium-dependent nature of the re-entrant circuit [15].
- RVOT/LVOT VT: Intravenous beta-blockers or adenosine are highly effective at terminating these cAMP-mediated triggered arrhythmias [11].
Note: Amiodarone is not recommended as a first-line agent for idiopathic VTs.

6. Management of Hemodynamically Unstable VT
Definition of Instability
Hemodynamic instability in the setting of VT is defined by the presence of one or more of the following: hypotension (typically systolic BP < 90 mmHg), altered mental status, signs of systemic shock (cool, clammy extremities, oliguria), ongoing ischemic chest pain, or acute pulmonary edema [40]. Unstable VT is an immediate threat to life.
Acute Management Approach
The management of unstable VT is dictated by the Advanced Cardiovascular Life Support (ACLS) guidelines and depends entirely on the presence or absence of a palpable pulse [40][41].
- Unstable VT with a Pulse:
- The definitive treatment is immediate synchronized electrical cardioversion. Synchronization to the R wave is crucial to avoid delivering a shock during the relative refractory period of the cardiac cycle (the T wave), which could precipitate ventricular fibrillation (the “R-on-T” phenomenon).
- If the patient is conscious and blood pressure permits, rapid administration of a short-acting sedative (e.g., midazolam or etomidate) is recommended to provide amnesia and analgesia. However, cardioversion must never be delayed if the patient is rapidly deteriorating.
- Initial energy selection is typically 100 Joules (biphasic), escalating to 200 Joules if unsuccessful [40].
- Pulseless VT:
- Pulseless VT is treated identically to ventricular fibrillation. It requires the immediate initiation of high-quality cardiopulmonary resuscitation (CPR) and unsynchronized defibrillation at maximum energy (typically 200 Joules biphasic) [40].
- Following the first shock, CPR is immediately resumed for 2 minutes before rhythm and pulse checks.
- If the rhythm remains shockable (refractory pulseless VT/VF), intravenous epinephrine (1 mg every 3-5 minutes) is administered.
- For VT/VF refractory to at least two shocks and epinephrine, antiarrhythmic therapy is indicated. Options include an intravenous amiodarone bolus (300 mg, followed by an additional 150 mg if needed) or intravenous lidocaine (1-1.5 mg/kg) [41].

7. Comprehensive Evaluation of the VT Patient
Following acute stabilization, every patient presenting with new-onset VT requires a meticulous evaluation to define the underlying etiology, assess the risk of recurrence, and guide long-term management [1].
Non-Invasive Evaluation
- 12-Lead ECG: Essential both during the tachycardia (to localize the exit site and suggest the mechanism) and during sinus rhythm. The sinus rhythm ECG can reveal underlying substrates: Q waves indicating prior MI, fragmented QRS complexes suggesting myocardial scar, prolonged QT intervals, Brugada patterns, or Epsilon waves [1].
- Echocardiography: The first-line imaging modality to assess global LVEF, identify regional wall motion abnormalities (suggestive of ischemia or scar), and evaluate valvular structural integrity.
- Cardiac Magnetic Resonance (CMR) Imaging: CMR has revolutionized VT risk stratification. The 2022 ESC Guidelines strongly recommend CMR (Class I) for patients with suspected NICM, ARVC, or HCM. Late Gadolinium Enhancement (LGE) is the gold standard for identifying the presence, extent, and precise anatomical distribution of myocardial scar and interstitial fibrosis. The presence and mass of LGE correlate directly with arrhythmogenic risk and are invaluable for pre-procedural planning, allowing electrophysiologists to target specific fibrotic channels during ablation [42][43].
Invasive and Specialized Testing
- Coronary Angiography: Indicated in patients with new-onset VT and intermediate-to-high pre-test probability of coronary artery disease to rule out acute ischemia as a reversible trigger.
- Electrophysiology Study (EPS): While not routinely required for all patients, EPS is highly useful for risk stratification in specific cohorts (e.g., patients with ARVC, or patients with previous MI and borderline LVEF 36-40%). It is also the definitive test for differentiating complex SVT with aberrancy from VT [1]. During EPS, voltage mapping correlates closely with CMR LGE, identifying areas of low voltage (<1.5 mV) that represent scar tissue.
- Genetic and Provocative Testing: Multidisciplinary genetic counseling and testing are recommended for patients with suspected channelopathies, ARVC, or familial cardiomyopathies. Provocative testing, such as the administration of sodium channel blockers (Ajmaline or Flecainide) to unmask a concealed Brugada pattern, or Epinephrine infusion to provoke CPVT, is utilized in specialized centers [8].

8. Long-Term Management
Long-term management aims to prevent sudden cardiac death, reduce VT recurrence, and minimize symptom burden. The strategy relies on a triad of device therapy, catheter ablation, and pharmacotherapy.
Implantable Cardioverter-Defibrillator (ICD) Therapy
The ICD is the most effective therapy for the prevention of SCD, terminating life-threatening arrhythmias via anti-tachycardia pacing (ATP) or high-voltage shocks.
- Secondary Prevention: ICD implantation is universally indicated (Class I) for survivors of SCD or hemodynamically unstable VT occurring in the absence of a reversible cause (such as acute STEMI or severe transient electrolyte derangement) [2].
- Primary Prevention: Indicated for patients at high risk of SCD who have not yet experienced a sustained ventricular arrhythmia.
- Ischemic Cardiomyopathy: Based on the landmark MADIT-II and SCD-HeFT trials, an ICD is indicated for patients with prior MI, LVEF ≤ 35%, and NYHA class II-III symptoms despite ≥ 3 months of optimal medical therapy (OMT) [44][45]. Sub-group analyses of MADIT-II demonstrated that patients with a QRS duration > 120 ms derived the most profound mortality benefit.
- Non-Ischemic Cardiomyopathy: The indication is more nuanced following the DANISH trial (2016), which showed that prophylactic ICD implantation in patients with NICM did not significantly reduce overall all-cause mortality compared to OMT alone [46]. However, critical sub-group analyses of the DANISH data revealed a significant mortality benefit in younger patients (age < 68 years). Consequently, primary prevention ICDs in NICM require individualized, shared decision-making, heavily weighing patient age, comorbidity burden, and the presence of extensive LGE on CMR [1].
Catheter Ablation: A Paradigm Shift to First-Line Therapy
Historically viewed as a palliative, last-resort option for patients failing multiple antiarrhythmic drugs, catheter ablation has rapidly ascended the treatment algorithms.
- Idiopathic VT: Catheter ablation is the first-line therapy (Class I) for symptomatic idiopathic VTs (RVOT, fascicular). It offers a definitive cure with success rates exceeding 90% and a very low complication profile [7].
- Ischemic VT (The VANISH Era): The management of ischemic VT has been revolutionized by recent randomized controlled trials.
- The VANISH trial (2016) demonstrated that in patients with ischemic cardiomyopathy and an ICD who experience VT despite initial amiodarone therapy, catheter ablation is significantly superior to escalating antiarrhythmic drug therapy in reducing the composite of death, VT storm, or appropriate ICD shocks [16].
- The landmark VANISH2 trial (presented at AHA 2024, published in NEJM) pushed the envelope further. It evaluated patients with prior MI, an ICD, and relatively well-tolerated VT who had not yet failed antiarrhythmic drugs. VANISH2 demonstrated that first-line catheter ablation is superior to first-line antiarrhythmic drugs (sotalol or amiodarone) in reducing the primary composite endpoint of death, VT storm, or appropriate ICD shocks [47]. This establishes ablation as the preferred initial strategy over drugs for ischemic VT.
- Similarly, the PAUSE-SCD trial (2022) and SURVIVE-VT trial demonstrated that early, first-line ablation at the time of ICD implantation significantly reduces VT burden, ICD shocks, and heart failure hospitalizations compared to conventional medical therapy [48][49].
Pharmacotherapy
While taking a secondary role to ablation for definitive control, pharmacotherapy remains essential.
- Beta-blockers: The foundation of therapy. They reduce sympathetic tone, decrease myocardial oxygen demand, and are the only antiarrhythmic class proven to reduce all-cause mortality in HFrEF [1].
- Amiodarone and Sotalol: Utilized to reduce VT burden and minimize painful ICD shocks in patients who are not candidates for, or have failed, catheter ablation. However, neither drug improves overall survival. Amiodarone, while highly effective, is plagued by significant long-term toxicities (pulmonary fibrosis, thyroid dysfunction, hepatotoxicity, and corneal microdeposits), necessitating rigorous surveillance [50][51].

9. Registry Data, Ongoing Trials, and Future Directions
Insights from Registry Data
Real-world registry data provide crucial insights that complement randomized trials. Data from the National Cardiovascular Data Registry (NCDR) ICD Registry highlight that despite optimal programming, up to 20% of primary prevention ICD recipients experience appropriate shocks within 3 years, underscoring the need for aggressive upstream substrate modification [52]. Furthermore, the International VT Ablation Center Collaborative Group (IVTCC) registry has demonstrated that while acute procedural success for VT ablation is high, long-term recurrence remains a challenge, particularly in non-ischemic cardiomyopathies where the arrhythmogenic substrate is often deep mid-wall or epicardial, making it difficult to reach with standard endocardial catheters [53].
Ongoing Trials and Innovations
The field of VT management is rapidly evolving, with several ongoing trials poised to further refine clinical practice:
- Pulsed Field Ablation (PFA): While currently revolutionizing atrial fibrillation management, PFA—a non-thermal energy source that causes cell death via electroporation—is under intense investigation for VT. Preclinical and early human feasibility studies suggest PFA can create deep, transmural lesions in ventricular myocardium while sparing adjacent structures like coronary arteries and the phrenic nerve. Ongoing trials are evaluating its safety and efficacy in ischemic and non-ischemic VT substrates [54].
- Non-Invasive Stereotactic Body Radiation Therapy (SBRT): For patients with refractory VT storm who are too hemodynamically unstable for conventional catheter ablation, cardiac radioablation (SBRT) is emerging as a salvage therapy. By delivering highly targeted, high-dose radiation to the arrhythmogenic scar (mapped non-invasively via ECG-imaging and CMR), SBRT induces localized fibrosis and conduction block. Following the promising results of the ENCORE-VT phase I/II trial, larger multicenter registries and randomized trials (e.g., the ADVANCE-VT trial) are currently assessing long-term safety and efficacy [55][56].
- PREVENT-VT and LESS-VT: These ongoing trials continue to explore the optimal timing of ablation, specifically investigating whether prophylactic ablation of the arrhythmogenic substrate in high-risk patients before their first clinical episode of VT can improve overall survival and prevent the development of heart failure [57].
In conclusion, the management of ventricular tachycardia has evolved into a highly sophisticated, multi-modality discipline. By integrating precise electrocardiographic diagnosis, advanced imaging, early catheter ablation, and judicious device therapy, cardiologists can significantly alter the natural history of this formidable disease, reducing mortality and improving the quality of life for patients at risk of sudden cardiac death.
References
- Zeppenfeld K, Tfelt-Hansen J, de Riva M, et al. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur Heart J. 2022;43(40):3997-4126. doi:10.1093/eurheartj/ehac262
- Al-Khatib SM, Stevenson WG, Ackerman MJ, et al. 2017 AHA/ACC/HRS Guideline for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death. Circulation. 2018;138(11):e272-e391. doi:10.1161/CIR.0000000000000549
- Priori SG, Blomström-Lundqvist C, Mazzanti A, et al. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur Heart J. 2015;36(41):2793-2867. doi:10.1093/eurheartj/ehv316
- Koplan BA, Stevenson WG. Ventricular tachycardia and sudden cardiac death. Mayo Clin Proc. 2009;84(3):289-297. doi:10.1016/S0025-6196(11)61149-X
- Josephson ME. Recurrent ventricular tachycardia. In: Clinical Cardiac Electrophysiology: Techniques and Interpretations. 4th ed. Lippincott Williams & Wilkins; 2008:446-641.
- Stevenson WG, Friedman PL, Kocovic D, et al. Radiofrequency catheter ablation of ventricular tachycardia after myocardial infarction. Circulation. 1998;98(4):308-314. doi:10.1161/01.cir.98.4.308
- Lerman BB. Mechanism, Diagnosis, and Treatment of Outflow Tract Tachycardia. Nat Rev Cardiol. 2015;12(10):597-608. doi:10.1038/nrcardio.2015.121
- Ackerman MJ, Priori SG, Willems S, et al. HRS/EHRA expert consensus statement on the state of genetic testing for the channelopathies and cardiomyopathies. Heart Rhythm. 2011;8(8):1308-1339. doi:10.1016/j.hrthm.2011.05.020
- Myerburg RJ, Kessler KM, Castellanos A. Sudden cardiac death: epidemiology, transient risk, and intervention assessment. Ann Intern Med. 1993;119(12):1187-1197. doi:10.7326/0003-4819-119-12-199312150-00006
- Tada H, Ito S, Naito S, et al. Idiopathic ventricular arrhythmia arising from the mitral annulus: a distinct subgroup of idiopathic ventricular arrhythmias. J Am Coll Cardiol. 2005;45(6):877-886. doi:10.1016/j.jacc.2004.12.025
- Callans DJ, Menz V, Schwartzman D, et al. Repetitive monomorphic tachycardia from the left ventricular outflow tract: electrocardiographic patterns consistent with a left ventricular site of origin. J Am Coll Cardiol. 1997;29(5):1023-1027. doi:10.1016/s0735-1097(97)00033-0
- Ouyang F, Fotuhi P, Ho SY, et al. Repetitive monomorphic ventricular tachycardia originating from the aortic sinus cusp: electrocardiographic characterization for guiding catheter ablation. J Am Coll Cardiol. 2002;39(3):500-508. doi:10.1016/s0735-1097(01)01767-3
- Belhassen B, Rotmensch HH, Laniado S. Response of recurrent sustained ventricular tachycardia to verapamil. Br Heart J. 1981;46(6):679-682. doi:10.1136/hrt.46.6.679
- Nogami A. Idiopathic left ventricular tachycardia: assessment and treatment. Card Electrophysiol Rev. 2002;6(4):448-457. doi:10.1023/a:1021132617757
- Zipes DP, Camm AJ, Borggrefe M, et al. ACC/AHA/ESC 2006 guidelines for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. J Am Coll Cardiol. 2006;48(5):e247-346. doi:10.1016/j.jacc.2006.07.010
- Sapp JL, Wells GA, Parkash R, et al. Antiarrhythmic Drug Therapy and Radiofrequency Catheter Ablation in Patients with Ischemic Ventricular Tachycardia (VANISH). N Engl J Med. 2016;375(2):111-121. doi:10.1056/NEJMoa1602045
- Marchlinski FE, Callans DJ, Gottlieb CD, Zado E. Linear ablation lesions for control of unmappable ventricular tachycardia in patients with ischemic and nonischemic cardiomyopathy. Circulation. 2000;101(11):1288-1296. doi:10.1161/01.cir.101.11.1288
- Maron BJ, Desai MY, Nishimura RA, et al. Diagnosis and Evaluation of Hypertrophic Cardiomyopathy: JACC State-of-the-Art Review. J Am Coll Cardiol. 2022;79(4):372-389. doi:10.1016/j.jacc.2021.12.002
- Corrado D, Link MS, Calkins H. Arrhythmogenic Right Ventricular Cardiomyopathy. N Engl J Med. 2017;376(1):61-72. doi:10.1056/NEJMra1509267
- Marcus FI, McKenna WJ, Sherrill D, et al. Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: proposed modification of the task force criteria. Circulation. 2010;121(13):1533-1541. doi:10.1161/CIRCULATIONAHA.108.840827
- Packer M. The neurohormonal hypothesis: a theory to explain the mechanism of disease progression in heart failure. J Am Coll Cardiol. 1992;20(1):248-254. doi:10.1016/0735-1097(92)90167-l
- Schwartz PJ, Crotti L, George AL Jr. Long QT Syndrome. Circ Arrhythm Electrophysiol. 2012;5(4):868-877. doi:10.1161/CIRCEP.111.962019
- Brugada P, Brugada J. Right bundle branch block, persistent ST segment elevation and sudden cardiac death: a distinct clinical and electrocardiographic syndrome. J Am Coll Cardiol. 1992;20(6):1391-1396. doi:10.1016/0735-1097(92)90253-j
- Leenhardt A, Lucet V, Denjoy I, et al. Catecholaminergic polymorphic ventricular tachycardia in children. A 7-year follow-up of 21 patients. Circulation. 1995;91(5):1512-1519. doi:10.1161/01.cir.91.5.1512
- Negru AG, et al. A Comprehensive Review of a Mechanism-Based Ventricular Electrical Storm Management. J Clin Med. 2025;14(15):5351. [Verify DOI]
- Gatzoulis KA, Andrikopoulos GK, Apostolopoulos T, et al. Electrical storm is an independent predictor of adverse long-term outcome in the era of implantable defibrillator therapy. Europace. 2005;7(2):184-192. doi:10.1016/j.eupc.2005.01.003
- Chyou J, et al. Management of Ventricular Tachycardia and Electrical Storm in the CICU. Cardionerds. 2023. [Verify Source]
- Chatzidou S, Kontogiannis C, Tsilimigras DI, et al. Propranolol versus metoprolol for treatment of electrical storm in patients with implantable cardioverter-defibrillator. J Am Coll Cardiol. 2018;71(17):1897-1906. doi:10.1016/j.jacc.2018.02.056
- Nademanee K, Taylor R, Bailey WE, et al. Treating electrical storm: sympathetic blockade versus advanced life support-guided therapy. Circulation. 2000;102(7):742-747. doi:10.1161/01.cir.102.7.742
- Bourke T, Vaseghi M, Michowitz Y, et al. Neuraxial modulation for refractory ventricular arrhythmias: value of thoracic epidural anesthesia and surgical left cardiac sympathetic denervation. Circulation. 2010;121(21):2255-2262. doi:10.1161/CIRCULATIONAHA.109.929703
- Carbucicchio C, Santamaria M, Trevisi N, et al. Catheter ablation for the treatment of electrical storm in patients with implantable cardioverter-defibrillators: short- and long-term outcomes in a prospective single-center study. Circulation. 2008;117(4):462-469. doi:10.1161/CIRCULATIONAHA.106.686535
- Wellens HJ, Bär FW, Lie KI. The value of the electrocardiogram in the differential diagnosis of a tachycardia with a widened QRS complex. Am J Med. 1978;64(1):27-33. doi:10.1016/0002-9343(78)90176-6
- Griffith MJ, de Belder MA, Linker NJ, et al. Multivariate analysis to simplify the differential diagnosis of broad complex tachycardia. Br Heart J. 1991;66(2):166-174. doi:10.1136/hrt.66.2.166
- Brugada P, Brugada J, Mont L, Smeets J, Andries EW. A new approach to the differential diagnosis of a regular tachycardia with a wide QRS complex. Circulation. 1991;83(5):1649-1659. doi:10.1161/01.cir.83.5.1649
- Vereckei A, Duray G, Szénási G, Altemose GT, Miller JM. New algorithm using only lead aVR for differential diagnosis of wide QRS complex tachycardia. Heart Rhythm. 2008;5(1):89-98. doi:10.1016/j.hrthm.2007.09.020
- Moccetti F, et al. The Basel Algorithm for the Differential Diagnosis of Wide QRS Tachycardias. JACC Clin Electrophysiol. 2022;8(7):846-857. doi:10.1016/j.jacep.2022.04.015
- LoCoco A, et al. Validation of the Basel Algorithm in Emergency Department settings. Am J Emerg Med. 2024. [Verify DOI]
- Ortiz M, Martín A, Arribas F, et al. Randomized comparison of intravenous procainamide vs. intravenous amiodarone for the acute treatment of tolerated wide QRS tachycardia: the PROCAMIO study. Eur Heart J. 2017;38(17):1329-1335. doi:10.1093/eurheartj/ehw027
- Marill KA, deSouza IS, Nishijima DK, et al. Amiodarone is poorly effective for the acute termination of ventricular tachycardia. Ann Emerg Med. 2006;47(3):217-224. doi:10.1016/j.annemergmed.2005.10.024
- Panchal AR, Bartos JA, Cabañas JG, et al. Part 3: Adult Basic and Advanced Life Support: 2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2020;142(16_suppl_2):S366-S468. doi:10.1161/CIR.0000000000000916
- Link MS, Berkow LC, Kudenchuk PJ, et al. Part 7: Adult Advanced Cardiovascular Life Support: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2015;132(18 Suppl 2):S444-S464. doi:10.1161/CIR.0000000000000261
- Hundley WG, Bluemke DA, Finn JP, et al. ACCF/ACR/AHA/NASCI/SCMR 2010 expert consensus document on cardiovascular magnetic resonance. J Am Coll Cardiol. 2010;55(23):2614-2662. doi:10.1016/j.jacc.2009.11.011
- Kramer CM, Barkhausen J, Flamm SD, Kim RJ, Nagel E. Standardized cardiovascular magnetic resonance imaging (CMR) protocols, society for cardiovascular magnetic resonance: board of trustees task force on standardized protocols. J Cardiovasc Magn Reson. 2013;15:91. doi:10.1186/1532-429X-15-91
- Moss AJ, Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction (MADIT-II). N Engl J Med. 2002;346(12):877-883. doi:10.1056/NEJMoa013474
- Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure (SCD-HeFT). N Engl J Med. 2005;352(3):225-237. doi:10.1056/NEJMoa043399
- Køber L, Thune JJ, Nielsen JC, et al. Defibrillator Implantation in Patients with Nonischemic Systolic Heart Failure (DANISH). N Engl J Med. 2016;375(13):1221-1230. doi:10.1056/NEJMoa1608029
- Sapp JL, et al. Catheter Ablation or Antiarrhythmic Drug Therapy for Ventricular Tachycardia in Ischemic Cardiomyopathy: The VANISH2 Trial. N Engl J Med. 2024. [Presented AHA 2024]. [Verify DOI]
- Tung R, Xue Y, Chen M, et al. First-Line Catheter Ablation of Monomorphic Ventricular Tachycardia in Cardiomyopathy Concurrent With Defibrillator Implantation: The PAUSE-SCD Randomized Trial. Circulation. 2022;145(25):1839-1849. doi:10.1161/CIRCULATIONAHA.122.060039
- Willems S, Tilz RR, Steven D, et al. Preventive or Deferred Ablation of Ventricular Tachycardia in Patients With Ischemic Cardiomyopathy and Implantable Defibrillator (BERLIN VT): A Multicenter Randomized Trial. Circulation. 2020;141(13):1057-1067. doi:10.1161/CIRCULATIONAHA.119.043400
- Connolly SJ, Dorian P, Roberts RS, et al. Comparison of beta-blockers, amiodarone plus beta-blockers, or sotalol for prevention of shocks from implantable cardioverter defibrillators: the OPTIC Study. JAMA. 2006;295(2):165-171. doi:10.1001/jama.295.2.165
- Sears SF, Todaro AW, Lewis TS, et al. Examining the impact of implantable cardioverter defibrillator shocks on patient outcomes. Clin Cardiol. 2018;41(1):130-136. doi:10.1002/clc.22836
- Hess PL, Laird A, Edwards R, et al. Survival and device-related complications in the National Cardiovascular Data Registry Implantable Cardioverter-Defibrillator Registry. J Am Coll Cardiol. 2014;64(11):1148-1158. doi:10.1016/j.jacc.2014.06.1186
- Tung R, Vaseghi M, Frankel DS, et al. Freedom from recurrent ventricular tachycardia after catheter ablation is associated with improved survival in patients with structural heart disease: An International VT Ablation Center Collaborative Group study. Heart Rhythm. 2015;12(9):1997-2007. doi:10.1016/j.hrthm.2015.05.036
- Reddy VY, Neuzil P, Koruth JS, et al. Pulsed Field Ablation for Ventricular Tachycardia. Circulation. 2022. [Verify DOI]
- Robinson CG, Samson PP, Moore KMS, et al. Phase I/II Trial of Electrophysiology-Guided Noninvasive Cardiac Radioablation for Ventricular Tachycardia. Circulation. 2019;139(3):313-321. doi:10.1161/CIRCULATIONAHA.118.038261
- Cuculich PS, Schill MR, Kashani R, et al. Noninvasive Cardiac Radiation for Ablation of Ventricular Tachycardia. N Engl J Med. 2017;377(24):2325-2336. doi:10.1056/NEJMoa1613773
- Ongoing Clinical Trials Registry (ClinicalTrials.gov). PREVENT-VT and LESS-VT study protocols. Accessed 2024.

