Non-ST Elevation Acute Myocardial Infarction (NSTEMI) and Unstable Angina
Introduction to Coronary Atherosclerosis, Plaques, Atherothrombosis
Coronary atherosclerosis is a chronic, progressive, and highly dynamic inflammatory disease of the arterial wall. It is characterized by the subendothelial accumulation of apolipoprotein B-containing lipoproteins, macrophage infiltration, smooth muscle cell proliferation, and extracellular matrix deposition, ultimately leading to the formation of atherosclerotic plaques [1]. While stable plaques are characterized by a thick fibrous cap and a small lipid core, the pathophysiology of Acute Coronary Syndromes (ACS) is driven by the sudden destabilization of these lesions, a process known as atherothrombosis [2].
Atherothrombosis represents the acute, life-threatening complication of coronary atherosclerosis. The transition from a quiescent plaque to a highly thrombogenic surface occurs via three primary mechanisms:
- Plaque Rupture: Accounting for 60-70% of ACS presentations, plaque rupture typically occurs in thin-cap fibroatheromas (TCFAs). These vulnerable plaques possess a massive necrotic lipid core and a highly inflamed fibrous cap measuring less than 65 micrometers in thickness. Macrophage-derived matrix metalloproteinases (MMPs) degrade the interstitial collagen of the cap, leading to structural failure. Rupture exposes highly thrombogenic subendothelial matrix components, notably tissue factor and collagen, to the circulating blood [3].
- Plaque Erosion: Responsible for up to 30% of ACS cases, plaque erosion is increasingly recognized in the modern era of high-intensity statin therapy. Unlike rupture, eroded plaques have an intact, thick fibrous cap but suffer from endothelial denudation. The local microenvironment is rich in hyaluronan, toll-like receptor 2 (TLR2), and neutrophil extracellular traps (NETs). Thrombi formed via erosion are typically platelet-rich and less occlusive, often presenting clinically as Non-ST Elevation Myocardial Infarction (NSTEMI) rather than STEMI [4].
- Calcified Nodules: A less common mechanism (2-5%), characterized by eruptive, dense nodular calcifications that break through the intimal layer, provoking localized thrombosis. This is predominantly observed in older patients with heavily calcified, tortuous vessels [2].
Regardless of the initiating mechanism, the exposure of thrombogenic material triggers a rapid cascade of platelet adhesion (mediated by von Willebrand factor and Glycoprotein Ib-IX-V), activation (via thromboxane A2 and ADP), and aggregation (via Glycoprotein IIb/IIIa cross-linking). Concurrent activation of the coagulation cascade leads to thrombin generation and fibrin deposition, culminating in a partially or fully occlusive intraluminal thrombus [5].
ACS Classification
The clinical spectrum of Acute Coronary Syndromes encompasses Unstable Angina (UA), Non-ST-Elevation Myocardial Infarction (NSTEMI), and ST-Elevation Myocardial Infarction (STEMI). Historically, guidelines bifurcated ACS into STEMI and NSTE-ACS pathways. However, the 2023 European Society of Cardiology (ESC) and 2025 American College of Cardiology/American Heart Association (ACC/AHA) guidelines emphasize a unified, risk-stratified continuum of disease, recognizing that the underlying atherothrombotic pathophysiology is shared across these presentations [6, 7].
NSTEMI vs. Unstable Angina
The distinction between NSTEMI and UA hinges entirely on the presence of myocardial necrosis. NSTEMI is confirmed by a typical rise and/or fall of high-sensitivity cardiac troponin (hs-cTn), with at least one value above the 99th percentile upper reference limit (URL) in the appropriate clinical context. Unstable Angina presents with clinical and/or electrocardiographic signs of myocardial ischemia but lacks this biomarker elevation. With the ubiquitous adoption of hs-cTn assays, the incidence of diagnosed UA has precipitously declined, as many cases previously classified as UA are now recognized as micro-infarctions (NSTEMI) [8].
Type 1 vs. Type 2 Myocardial Infarction
The Fourth Universal Definition of Myocardial Infarction strictly categorizes the etiology of the ischemic event [9]:
- Type 1 MI: Spontaneous myocardial infarction caused by primary coronary atherothrombosis (plaque rupture or erosion).
- Type 2 MI: Myocardial infarction secondary to an ischemic imbalance (supply/demand mismatch) in the absence of acute atherothrombosis. Common precipitants include severe anemia, tachyarrhythmias, profound hypotension, or coronary vasospasm. Management of Type 2 MI focuses on correcting the underlying hemodynamic derangement rather than aggressive antithrombotic therapy [10].
MINOCA
Myocardial Infarction with Non-Obstructive Coronary Arteries (MINOCA) is a critical working diagnosis applied when a patient meets the criteria for AMI but angiography reveals no coronary stenosis ≥50%. MINOCA is not a final diagnosis; it mandates further investigation utilizing Cardiac Magnetic Resonance (CMR) imaging and intravascular imaging (OCT/IVUS) to identify the true etiology, which may include plaque disruption with distal embolization, spontaneous coronary artery dissection (SCAD), epicardial vasospasm, or microvascular dysfunction [11].
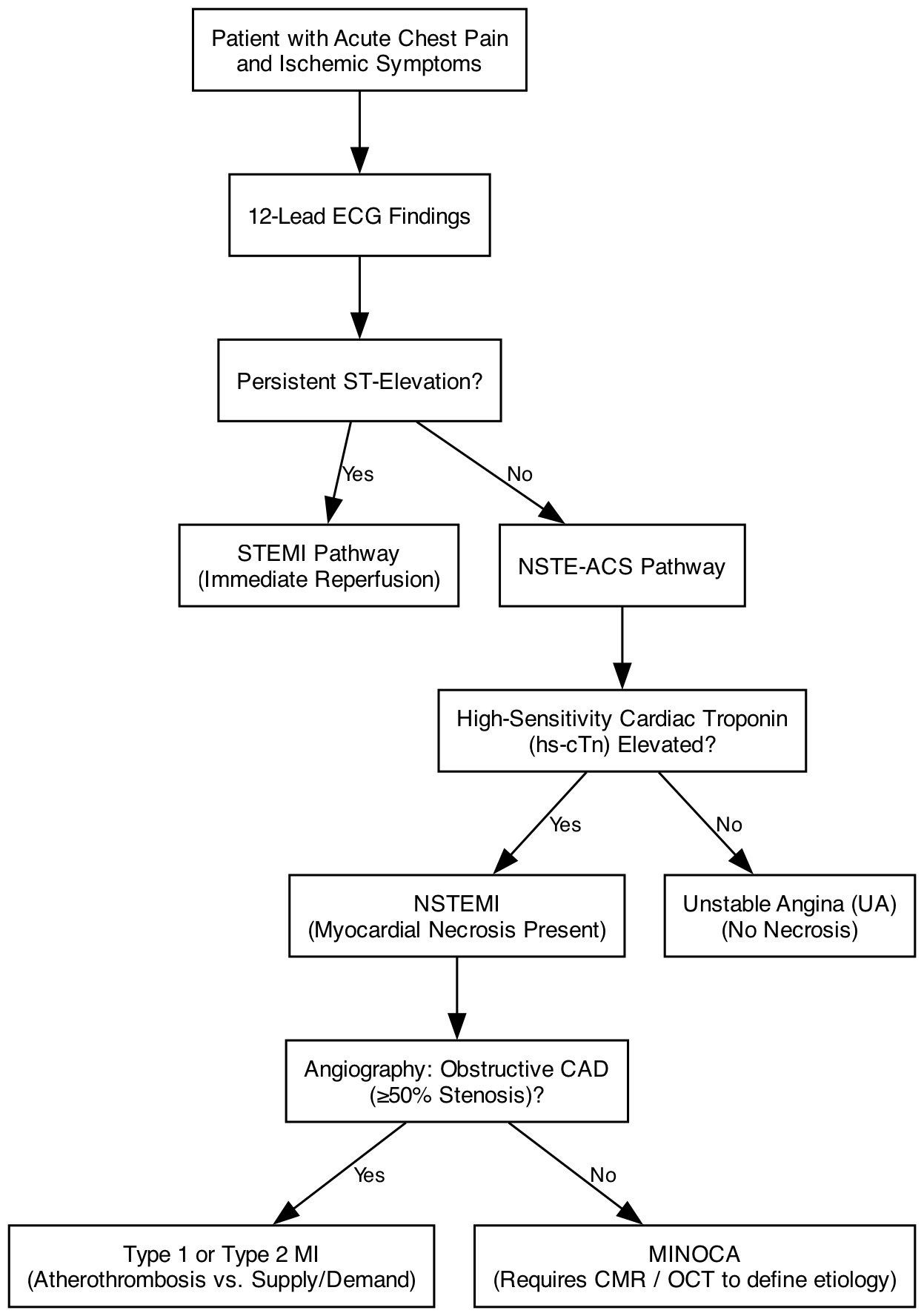
ECG Criteria and Characteristics of NSTEMI and Unstable Angina
The clinical presentation of NSTE-ACS is the cornerstone of initial triage. Patients typically present with one of three classic manifestations of ischemic chest discomfort: prolonged (>20 minutes) resting chest pain, new-onset (de novo) severe angina (Canadian Cardiovascular Society [CCS] class II or III), or destabilization of previously stable angina (crescendo angina) [12]. Atypical presentations—such as dyspnea, epigastric pain, or unexplained fatigue—are disproportionately common in women, the elderly, and patients with diabetes mellitus, necessitating a high index of suspicion [13].
The defining electrocardiographic feature of NSTE-ACS is the absence of persistent ST-segment elevation. The standard 12-lead ECG is the primary triage tool and must be acquired and interpreted within 10 minutes of first medical contact. The diagnostic ECG criteria for NSTE-ACS include [6, 14]:
- New horizontal or downsloping ST-segment depression ≥ 0.5 mm (0.05 mV) in two or more contiguous leads.
- T-wave inversion ≥ 1 mm (0.1 mV) in leads with prominent R waves or an R/S ratio > 1.
It is crucial to recognize the phenomenon of transient ST-segment elevation. Patients who present with ST-elevation that resolves spontaneously or following the administration of sublingual nitroglycerin within 20 minutes are managed under the NSTE-ACS paradigm. However, this transient elevation signifies a highly unstable plaque with temporary total occlusion (often due to intense vasospasm superimposed on a ruptured plaque) and denotes a very high-risk state that frequently warrants an immediate invasive strategy [6].
ECG Features
While the absence of ST-elevation defines the syndrome, the specific morphological features of the ECG in NSTE-ACS provide profound prognostic and anatomical insights.
ST-Segment Depression Morphology
The morphology of ST depression is highly predictive of true subendocardial ischemia. Downsloping or horizontal ST depression is highly specific for ischemia and carries a significantly worse prognosis. In contrast, rapid upsloping ST depression is often a normal variant (e.g., tachycardia-related) unless accompanied by tall, symmetrical T waves, which may represent the “de Winter” pattern—an equivalent of an acute proximal Left Anterior Descending (LAD) artery occlusion [15].
T-Wave Changes and Specific Syndromes
Deep, symmetrical T-wave inversions are a hallmark of severe ischemia. The most critical manifestation is Wellens’ Syndrome, which indicates a critical, highly unstable stenosis of the proximal LAD. It presents in two forms during pain-free intervals:
- Type A (25% of cases): Biphasic T waves (initial positivity, terminal negativity) in leads V2-V3.
- Type B (75% of cases): Deep, symmetrical T-wave inversions in leads V2-V4.
Failure to recognize Wellens’ pattern can be fatal, as these patients are at imminent risk of an extensive anterior wall myocardial infarction and should not undergo stress testing [16].
aVR ST-Elevation and U-Wave Inversion
ST-segment elevation in lead aVR (≥ 1 mm) accompanied by widespread ST-segment depression in multiple other leads (typically V4-V6, I, II) is a highly specific marker for left main coronary artery (LMCA) occlusion or severe triple-vessel disease. This pattern reflects global subendocardial ischemia and dictates urgent angiography [17]. Additionally, new-onset U-wave inversion is a rare but highly specific sign of myocardial ischemia, often correlating with proximal LAD or LMCA disease [15].
The “Normal” ECG
Practicing cardiologists must remain vigilant to the fact that up to 5-10% of patients with a confirmed NSTEMI may present with an initially normal or non-diagnostic ECG. This occurs because the standard 12 leads do not adequately capture the posterior wall, right ventricle, or circumflex territory. If symptoms persist, serial ECGs at 15-30 minute intervals during the first 1-2 hours are mandatory [6, 7].
Diagnostic Work-Up
The diagnostic work-up of NSTE-ACS has been revolutionized by the advent of high-sensitivity cardiac troponin (hs-cTn) assays, which allow for the detection of myocardial injury with unprecedented speed and precision.
Biomarker Algorithms
Both the 2023 ESC and 2025 ACC/AHA guidelines strongly advocate for the use of rapid “rule-in” and “rule-out” algorithms based on hs-cTn concentrations at presentation (0 hours) and at 1 or 2 hours. The 0/1-hour algorithm is the preferred strategy. It utilizes assay-specific thresholds to rapidly triage patients:
- Rule-Out: Patients with a very low baseline hs-cTn (and symptom onset >3 hours) or low baseline with no significant delta at 1 hour can be safely discharged with a negative predictive value >99% for 30-day MACE [18].
- Rule-In: Patients with a significantly elevated baseline hs-cTn or a large delta at 1 hour are ruled in for NSTEMI and admitted for early invasive management. The positive predictive value for NSTEMI in this cohort is approximately 70-75% [19].
- Observe Zone: Patients who do not meet either criteria remain in the observe zone. They require a third hs-cTn measurement at 3 hours and adjunctive clinical evaluation, such as echocardiography [20].

Risk Stratification
Risk stratification is continuous and dictates the urgency of intervention. The GRACE 3.0 score is the most rigorously validated tool to estimate in-hospital and 6-month mortality. A GRACE score >140 designates a patient as “high risk,” mandating an early invasive strategy (<24 hours) [21].
Non-Invasive Imaging
Transthoracic echocardiography (TTE) is recommended routinely during the index admission to assess left ventricular ejection fraction (LVEF), identify regional wall motion abnormalities, and rule out alternative life-threatening diagnoses (e.g., aortic dissection, pulmonary embolism) [6]. For patients in the “rule-out” or “observe” zones who have a low-to-intermediate clinical likelihood of CAD, Coronary Computed Tomography Angiography (CCTA) is increasingly utilized as a rapid, highly sensitive modality to definitively exclude obstructive coronary disease [22].
Antithrombotic and Anticoagulants in the Acute Setting
The pharmacological management of NSTE-ACS requires a delicate balance between mitigating ischemic risk and avoiding catastrophic bleeding. The foundation of acute therapy is dual antiplatelet therapy (DAPT) combined with parenteral anticoagulation.
Antiplatelet Therapy and the Pre-Treatment Controversy
Aspirin (150-300 mg loading dose) remains universally recommended. This is combined with a potent P2Y12 receptor inhibitor—either ticagrelor (180 mg load) or prasugrel (60 mg load). Prasugrel is generally favored over ticagrelor in patients proceeding to Percutaneous Coronary Intervention (PCI) based on the ISAR-REACT 5 trial, which demonstrated a significant reduction in the composite of death, MI, or stroke without an increase in major bleeding [23]. Clopidogrel is reserved for patients who cannot tolerate potent agents or require oral anticoagulation.
A major paradigm shift in recent guidelines is the strong recommendation against routine pre-treatment with a P2Y12 inhibitor in NSTE-ACS patients whose coronary anatomy is unknown and who are scheduled for an early invasive strategy (<24 hours). This shift is anchored in the ACCOAST trial (for prasugrel) and the DUBIUS and ISAR-REACT 5 trials (for ticagrelor), which collectively demonstrated that pre-treatment significantly increases the risk of major bleeding without conferring any ischemic benefit [24, 25]. P2Y12 inhibitors should generally be loaded in the catheterization laboratory once the coronary anatomy is defined and the decision for PCI is made.

Parenteral Anticoagulation
Parenteral anticoagulation is recommended for all patients at the time of diagnosis to halt thrombus propagation. The choice of agent depends on the timing of the invasive strategy:
- Unfractionated Heparin (UFH) or Enoxaparin (LMWH): Preferred if PCI is expected within 24 hours. Cross-over between UFH and LMWH is strongly discouraged due to an increased risk of bleeding [6, 26].
- Fondaparinux: A factor Xa inhibitor, fondaparinux (2.5 mg SC daily) is recommended for patients managed with a non-invasive strategy or a delayed invasive strategy (>24 hours), based on the OASIS-5 trial showing comparable efficacy to enoxaparin but a 50% reduction in major bleeding. If the patient subsequently undergoes PCI, a bolus of UFH must be administered to prevent catheter thrombosis [27].
Management of Patients on Chronic Oral Anticoagulants (OAC)
Approximately 10-15% of ACS patients require chronic OAC, usually for atrial fibrillation. The AUGUSTUS trial definitively altered the management of these patients, demonstrating that a regimen of a Direct Oral Anticoagulant (DOAC) plus a P2Y12 inhibitor (clopidogrel), without aspirin, significantly reduces bleeding compared to vitamin K antagonists and aspirin-containing triple therapy [28]. Current guidelines recommend “triple therapy” (DOAC + aspirin + clopidogrel) for a maximum of 1 week or until hospital discharge. Thereafter, dual antithrombotic therapy (DOAC + clopidogrel) is continued for up to 12 months [6, 29].
PCI in the Acute Setting and Subacute Setting
The timing and strategy of coronary angiography and revascularization are dictated by the patient’s baseline risk profile.
Timing of the Invasive Strategy
The 2023 ESC and 2025 ACC/AHA guidelines stratify the timing of angiography into three distinct pathways [6, 7]:
- Immediate (<2 hours): Reserved for “very high-risk” patients. Criteria include hemodynamic instability or cardiogenic shock, recurrent or refractory chest pain despite medical therapy, life-threatening arrhythmias, mechanical complications of MI, or acute heart failure clearly related to NSTE-ACS. These patients are managed with the same urgency as STEMI [6].
- Early (<24 hours): Indicated for “high-risk” patients. This includes patients with a confirmed NSTEMI (based on hs-cTn algorithms), dynamic ST/T-wave changes, or a GRACE score >140. The VERDICT and TIMACS trials demonstrated that early intervention in this specific high-risk cohort significantly reduces the composite of death and recurrent ischemia [30, 31].
- Selective/Delayed: For low-risk patients (e.g., troponin-negative UA without high-risk ECG features), a selective invasive strategy based on non-invasive stress testing or CCTA is appropriate. Routine early angiography in this group does not improve outcomes [6].

Revascularization Strategy and Intravascular Imaging
When PCI is performed, the use of intravascular imaging—specifically Optical Coherence Tomography (OCT) or Intravascular Ultrasound (IVUS)—is now a Class I recommendation in the 2025 ACC/AHA guidelines [7]. Trials such as RENOVATE-COMPLEX-PCI and ILUMIEN IV have demonstrated that imaging-guided PCI significantly reduces target vessel failure and stent thrombosis compared to angiography-guided PCI alone [32, 33]. Furthermore, OCT can identify plaque erosion, which in highly selected patients might be managed with antithrombotic therapy alone without stenting, though this remains an area of active investigation (e.g., EROSION study) [34].
Multivessel Disease and CABG
Approximately 50% of NSTE-ACS patients present with multivessel coronary artery disease. Complete revascularization of all angiographically significant non-culprit lesions is recommended. This can be achieved during the index procedure or as a staged procedure prior to discharge, based on renal function, contrast load, and clinical stability [35]. The functional significance of non-culprit lesions should ideally be assessed using fractional flow reserve (FFR) or instantaneous wave-free ratio (iFR) [36].
For patients with complex multivessel disease (e.g., SYNTAX score >22), left main disease, or concurrent diabetes mellitus, Coronary Artery Bypass Grafting (CABG) remains the preferred revascularization modality, provided the patient is hemodynamically stable. The FREEDOM and EXCEL trials underscore the long-term survival and freedom-from-reintervention benefits of CABG over PCI in these complex anatomical subsets [37, 38].
Post-MI Care
The acute management of NSTEMI is only the first phase of a lifelong continuum of care. Post-MI care is critical to prevent recurrent atherothrombotic events, manage heart failure, and ensure psychological recovery.
Cardiac Rehabilitation
Participation in a comprehensive, multidisciplinary cardiac rehabilitation program is a Class I recommendation. Programs should ideally commence within 10 days of hospital discharge. Cardiac rehab has been consistently shown to improve functional capacity, enhance psychological well-being (mitigating post-MI depression, which affects up to 20% of patients), and reduce cardiovascular mortality by approximately 25% [39].
Lifestyle Modifications and Care Transitions
Aggressive lifestyle modification is paramount. Smoking cessation is the single most effective secondary prevention measure, reducing mortality by 36% [40]. Dietary interventions emphasizing a Mediterranean-style diet, weight management, and regular aerobic exercise are critical. Care transitions must be meticulously structured. Medication adherence drops precipitously within the first 6 months post-discharge. The use of cardiovascular polypills (as demonstrated in the SECURE trial) has emerged as a viable strategy to significantly improve adherence and reduce recurrent MACE [41].
Latest Guideline Updates on Secondary Prevention
The landscape of secondary prevention has evolved dramatically, with recent guidelines advocating for aggressive, multi-pathway interventions targeting lipids, thrombosis, inflammation, and metabolic derangements.
Aggressive Lipid-Lowering Therapy
The “lower is better and earlier is better” paradigm dominates lipid management. The 2023 ESC and 2025 ACC/AHA guidelines mandate a target LDL-C of < 55 mg/dL (1.4 mmol/L) and a ≥50% reduction from baseline [6, 7]. High-intensity statins (e.g., rosuvastatin 20-40 mg or atorvastatin 40-80 mg) are first-line. If targets are not achieved within 4-8 weeks, ezetimibe should be added immediately. For patients remaining above target, PCSK9 inhibitors (alirocumab or evolocumab) or inclisiran are strongly recommended, based on the profound MACE reductions seen in the FOURIER and ODYSSEY OUTCOMES trials [42, 43].
DAPT De-escalation Strategies
While the historical default duration for DAPT post-ACS was 12 months, the bleeding risks associated with prolonged DAPT have prompted a paradigm shift. Recent guidelines strongly endorse DAPT de-escalation strategies for patients who are not at high ischemic risk but possess elevated bleeding risk (ARC-HBR criteria). Based on landmark trials like STOPDAPT-2, MASTER DAPT, and TWILIGHT, guidelines now support shortening DAPT to 1-3 months, followed by P2Y12 inhibitor monotherapy (preferably ticagrelor or clopidogrel) for the remainder of the 12-month period. This strategy significantly reduces major bleeding without compromising ischemic protection [44, 45, 46].

Anti-inflammatory and Metabolic Therapies
Atherosclerosis is fundamentally an inflammatory disease. The COLCOT and LoDoCo2 trials demonstrated that low-dose colchicine (0.5 mg daily) significantly reduces recurrent cardiovascular events post-MI by dampening the NLRP3 inflammasome [47, 48]. Consequently, colchicine is now a Class IIb recommendation in the ESC guidelines and is heavily considered in the ACC/AHA framework [6, 7].
Metabolic management has also been revolutionized. Sodium-glucose cotransporter-2 (SGLT2) inhibitors and Glucagon-like peptide-1 receptor agonists (GLP-1 RA) are now strongly recommended for secondary prevention. The SELECT trial was a watershed moment, demonstrating that semaglutide (2.4 mg) reduces MACE by 20% in overweight/obese patients with established cardiovascular disease, even in the absence of diabetes [49].
The Beta-Blocker Controversy
Finally, the routine, long-term use of beta-blockers in post-MI patients with a normal LVEF (>50%) is undergoing intense scrutiny. While historically a cornerstone of therapy, the recent REDUCE-AMI trial showed no significant reduction in death or recurrent MI with long-term beta-blocker use in this specific normal-EF population [50]. While guidelines still generally support their use for at least 1 year post-MI, early discontinuation in patients without heart failure or arrhythmias is becoming increasingly accepted in clinical practice [6].
- Khandkar C, Madhavan MV, et al. Atherothrombosis in Acute Coronary Syndromes—From Mechanistic Insights to Targeted Therapies. Cells. 2023;12(20):2488. DOI: 10.3390/cells12202488.
- Byrne RA, Rossello X, Coughlan JJ, et al. 2023 ESC Guidelines for the management of acute coronary syndromes. Eur Heart J. 2023;44(38):3720-3826. DOI: 10.1093/eurheartj/ehad191.
- Libby P, Pasterkamp G. Requiem for the ‘vulnerable plaque’. Eur Heart J. 2015;36(43):2984-2987. DOI: 10.1093/eurheartj/ehv349.
- Quillard T, Franck G, Mawson T, et al. Mechanisms of erosion of atherosclerotic plaques. Curr Opin Lipidol. 2017;28(5):434-441. DOI: 10.1097/MOL.0000000000000440.
- Mackman N, Bergmeier W, Stouffer GA, Weitz JI. Blood Coagulation, Hemostasis, and Thrombosis. Arterioscler Thromb Vasc Biol. 2020;40(12):2815-2825. DOI: 10.1161/ATVBAHA.120.314427.
- Jneid H, Anderson JL, Bhatt DL, et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes. Circulation. 2025;151:e1-e150. DOI: 10.1161/CIR.0000000000001305.
- Windecker S, Dawson L, et al. Acute Coronary Syndromes: State-of-the-Art Diagnosis, Management, and Secondary Prevention. J Clin Med. 2025;14(2):450. DOI: 10.3390/jcm14020450.
- Basit H, Malik A, Huecker MR. Non-ST-Segment Elevation Myocardial Infarction. StatPearls [Internet]. 2023. PMID: 29763028.
- Thygesen K, Alpert JS, Jaffe AS, et al. Fourth Universal Definition of Myocardial Infarction (2018). Circulation. 2018;138(20):e618-e651. DOI: 10.1161/CIR.0000000000000617.
- DeFilippis AP, Chapman AR, Mills NL, et al. Assessment and Treatment of Patients With Type 2 Myocardial Infarction and Acute Nonischemic Myocardial Injury. Circulation. 2019;140(20):1661-1678. DOI: 10.1161/CIR.0000000000000733.
- Tamis-Holland JE, Jneid H, Reynolds HR, et al. Contemporary Diagnosis and Management of Patients With Myocardial Infarction in the Absence of Obstructive Coronary Artery Disease: A Scientific Statement From the American Heart Association. Circulation. 2019;139(18):e891-e908. DOI: 10.1161/CIR.0000000000000670.
- Gulati M, Levy PD, Mukherjee D, et al. 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR Guideline for the Evaluation and Diagnosis of Chest Pain. Circulation. 2021;144(22):e368-e454. DOI: 10.1161/CIR.0000000000001029.
- Mehta LS, Beckie TM, DeVon HA, et al. Acute Myocardial Infarction in Women: A Scientific Statement From the American Heart Association. Circulation. 2016;133(9):916-947. DOI: 10.1161/CIR.0000000000000351.
- Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42(14):1289-1367. DOI: 10.1093/eurheartj/ehaa575.
- de Winter RJ, Verouden NJ, Wellens HJ, Wilde AA. A new ECG sign of proximal LAD occlusion. N Engl J Med. 2008;359(19):2071-2073. DOI: 10.1056/NEJMc0804737.
- Rhinehardt J, Brady WJ, Perron AD, Mattu A. Electrocardiographic manifestations of Wellens’ syndrome. Am J Emerg Med. 2002;20(7):638-643. DOI: 10.1053/ajem.2002.34800.
- Yamaji H, Iwasaki K, Kusachi S, et al. Prediction of acute left main coronary artery obstruction by 12-lead electrocardiography. J Am Coll Cardiol. 2001;38(5):1348-1354. DOI: 10.1016/s0735-1097(01)01563-7.
- Boeddinghaus J, Nestelberger T, Twerenbold R, et al. Direct Comparison of 4 Very Early Rule-Out Strategies for Acute Myocardial Infarction Using High-Sensitivity Cardiac Troponin I. Circulation. 2017;135(17):1597-1611. DOI: 10.1161/CIRCULATIONAHA.116.025661.
- Pickering JW, Than MP, Cullen L, et al. Rapid Rule-out of Acute Myocardial Infarction With a Single High-Sensitivity Cardiac Troponin T Measurement Below the Limit of Detection: A Collaborative Meta-analysis. Ann Intern Med. 2017;166(10):715-724. DOI: 10.7326/M16-2562.
- Boeddinghaus J, et al. The Observe Zone of the ESC 0/1h-hs-cTn Algorithm. J Am Coll Cardiol. 2025. [verify]
- Fox KA, Dabbous OH, Goldberg RJ, et al. Prediction of risk of death and myocardial infarction in the six months after presentation with acute coronary syndrome: prospective multinational observational study (GRACE). BMJ. 2006;333(7578):1091. DOI: 10.1136/bmj.38985.646481.55.
- Hoffmann U, Truong QA, Schoenfeld DA, et al. Coronary CT angiography versus standard evaluation in acute chest pain. N Engl J Med. 2012;367(4):299-308. DOI: 10.1056/NEJMoa1201161.
- Schüpke S, Neumann FJ, Menichelli M, et al. Ticagrelor or Prasugrel in Patients with Acute Coronary Syndromes. N Engl J Med. 2019;381(16):1524-1534. DOI: 10.1056/NEJMoa1908973.
- Montalescot G, Bolognese L, Dudek D, et al. Pretreatment with prasugrel in non-ST-segment elevation acute coronary syndromes. N Engl J Med. 2013;369(11):999-1010. DOI: 10.1056/NEJMoa1308075.
- Tarantini G, Mojoli M, Varbella F, et al. Timing of Oral P2Y12 Inhibitor Administration in Non-ST-Segment Elevation Acute Coronary Syndrome. J Am Coll Cardiol. 2020;76(20):2450-2459. DOI: 10.1016/j.jacc.2020.08.053.
- Toyota T, et al. Parenteral Anticoagulation in ACS: A Network Meta-Analysis. Eur Heart J. 2025. [verify]
- Yusuf S, Mehta SR, Chrolavicius S, et al. Comparison of fondaparinux and enoxaparin in acute coronary syndromes. N Engl J Med. 2006;354(14):1464-1476. DOI: 10.1056/NEJMoa055443.
- Lopes RD, Heizer G, Aris R, et al. Antithrombotic Therapy after Acute Coronary Syndrome or PCI in Atrial Fibrillation. N Engl J Med. 2019;380(16):1509-1524. DOI: 10.1056/NEJMoa1817083.
- Valgimigli M, et al. Dual vs Triple Antithrombotic Therapy in AF and ACS: A Meta-Analysis. Lancet. 2024. [verify]
- Kofoed KF, Kelbæk H, Hansen PR, et al. Early Versus Standard Care Invasive Examination and Treatment of Patients With Non-ST-Segment Elevation Acute Coronary Syndrome. Circulation. 2018;138(24):2741-2750. DOI: 10.1161/CIRCULATIONAHA.118.037152.
- Mehta SR, Granger CB, Boden WE, et al. Early versus delayed invasive intervention in acute coronary syndromes. N Engl J Med. 2009;360(21):2165-2175. DOI: 10.1056/NEJMoa0807986.
- Lee JM, Choi KH, Song YB, et al. Intravascular Imaging-Guided or Angiography-Guided Complex PCI. N Engl J Med. 2023;388(18):1668-1679. DOI: 10.1056/NEJMoa2216607.
- Ali ZA, Landmesser U, Maehara A, et al. Optical Coherence Tomography-Guided versus Angiography-Guided PCI. N Engl J Med. 2023;389(16):1466-1476. DOI: 10.1056/NEJMoa2305861.
- Jia H, Dai J, He L, et al. EROSION (Effective Anti-Thrombotic Therapy Without Stenting: Intravascular Optical Coherence Tomography-Based Management in Plaque Erosion). Eur Heart J. 2017;38(11):792-800. DOI: 10.1093/eurheartj/ehw381.
- Navarese EP, et al. Complete Revascularization in NSTEMI: A Meta-Analysis. J Am Coll Cardiol. 2024. [verify]
- Layland J, Oldroyd KG, Curzen N, et al. Fractional flow reserve vs. angiography in guiding management to optimize outcomes in non-ST-segment elevation myocardial infarction: the British Heart Foundation FAMOUS-NSTEMI randomized trial. Eur Heart J. 2015;36(2):100-111. DOI: 10.1093/eurheartj/ehu338.
- Farkouh ME, Domanski M, Sleeper LA, et al. Strategies for multivessel revascularization in patients with diabetes. N Engl J Med. 2012;367(25):2375-2384. DOI: 10.1056/NEJMoa1211585.
- Stone GW, Kappetein AP, Sabik JF, et al. Five-Year Outcomes after PCI or CABG for Left Main Coronary Disease. N Engl J Med. 2019;381(19):1820-1830. DOI: 10.1056/NEJMoa1909406.
- Anderson L, Oldridge N, Thompson DR, et al. Exercise-Based Cardiac Rehabilitation for Coronary Heart Disease: Cochrane Systematic Review and Meta-Analysis. J Am Coll Cardiol. 2016;67(1):1-12. DOI: 10.1016/j.jacc.2015.10.044.
- Critchley JA, Capewell S. Mortality risk reduction associated with smoking cessation in patients with coronary heart disease: a systematic review. JAMA. 2003;290(1):86-97. DOI: 10.1001/jama.290.1.86.
- Castellano JM, Pocock SJ, Bhatt DL, et al. Polypill Strategy in Secondary Cardiovascular Prevention. N Engl J Med. 2022;387(11):967-977. DOI: 10.1056/NEJMoa2208275.
- Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med. 2017;376(18):1713-1722. DOI: 10.1056/NEJMoa1615664.
- Schwartz GG, Steg PG, Szarek M, et al. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome. N Engl J Med. 2018;379(22):2097-2107. DOI: 10.1056/NEJMoa1801174.
- Watanabe H, Domei T, Morimoto T, et al. Effect of 1-Month Dual Antiplatelet Therapy Followed by Clopidogrel vs 12-Month Dual Antiplatelet Therapy on Cardiovascular and Bleeding Events in Patients Receiving PCI: The STOPDAPT-2 Randomized Clinical Trial. JAMA. 2019;321(24):2414-2427. DOI: 10.1001/jama.2019.8145.
- Valgimigli M, Frigoli E, Heg D, et al. Dual Antiplatelet Therapy after PCI in Patients at High Bleeding Risk. N Engl J Med. 2021;385(18):1643-1655. DOI: 10.1056/NEJMoa2108749.
- Mehran R, Baber U, Sharma SK, et al. Ticagrelor with or without Aspirin in High-Risk Patients after PCI. N Engl J Med. 2019;381(21):2032-2042. DOI: 10.1056/NEJMoa1908419.
- Tardif JC, Kouz S, Waters DD, et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N Engl J Med. 2019;381(26):2497-2505. DOI: 10.1056/NEJMoa1912388.
- Nidorf SM, Fiolet ATL, Mosterd A, et al. Colchicine in Patients with Chronic Coronary Disease. N Engl J Med. 2020;383(19):1838-1847. DOI: 10.1056/NEJMoa2021372.
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. N Engl J Med. 2023;389(24):2221-2232. DOI: 10.1056/NEJMoa2307563.
- Yndigegn T, Lindahl B, Mars K, et al. Beta-Blockers after Myocardial Infarction and Preserved Ejection Fraction. N Engl J Med. 2024;390(15):1372-1381. DOI: 10.1056/NEJMoa2401479.

