Heart Failure with Preserved Ejection Fraction (HFpEF)
Definition and terminology
Heart failure with preserved ejection fraction (HFpEF) is a complex clinical syndrome characterized by the cardinal symptoms and signs of heart failure (HF) in the presence of a left ventricular ejection fraction (LVEF) of ≥50%, accompanied by objective evidence of structural and/or functional cardiac abnormalities and/or elevated natriuretic peptides (NPs) [1][2]. The evolution of HF terminology has been driven by a deeper understanding of myocardial mechanics and clinical outcomes. The 2022 American Heart Association/American College of Cardiology/Heart Failure Society of America (AHA/ACC/HFSA) guidelines and the 2023 European Society of Cardiology (ESC) guidelines have universally solidified the LVEF categories: HF with reduced EF (HFrEF, ≤40%), HF with mildly reduced EF (HFmrEF, 41–49%), and HFpEF (≥50%) [1][2].
Crucially, contemporary definitions distinguish HFpEF from “Heart Failure with improved EF” (HFimpEF). HFimpEF refers to patients with a historical baseline LVEF ≤40% that has subsequently improved to >40% under guideline-directed medical therapy (GDMT). These patients must be managed as having HFrEF, as their underlying pathophysiology, trajectory, and risk of relapse upon GDMT withdrawal are fundamentally distinct from true, incident HFpEF [3]. Hemodynamically, the gold standard definition of HFpEF relies on the demonstration of spontaneous or provokable elevated left ventricular (LV) filling pressures, defined invasively as a pulmonary capillary wedge pressure (PCWP) ≥15 mmHg at rest or ≥25 mmHg during exercise [4]. This hemodynamic criterion underscores that “preserved” ejection fraction does not equate to normal cardiac function, but rather to a state where stroke volume is maintained only at the expense of pathologically elevated filling pressures.
Epidemiology and risk factors
HFpEF has reached epidemic proportions, now accounting for over 50% of all incident heart failure cases globally [5]. Registry data, including the Get With The Guidelines-Heart Failure (GWTG-HF) and the SWEDEHEART registries, demonstrate that while the incidence of HFrEF has stabilized or slightly declined over the past two decades, the incidence of HFpEF continues to rise steeply [6]. This epidemiological shift is primarily driven by the aging of the global population and the escalating prevalence of cardiometabolic comorbidities.
The demographic profile of the typical HFpEF patient differs markedly from that of HFrEF. Patients are predominantly older (frequently >65 years of age) and there is a strong female predominance. Women have a substantially higher lifetime risk of developing HFpEF, a phenomenon attributed to intrinsic sex differences in cardiovascular aging, including smaller ventricular cavities, greater age-related vascular stiffening, and distinct microvascular responses to systemic inflammation [7]. The dominant risk factors for HFpEF include hypertension (present in >80% of cases), obesity, type 2 diabetes mellitus, chronic kidney disease (CKD), sedentary lifestyle, and atrial fibrillation (AF) [5]. Unlike HFrEF, where epicardial ischemic heart disease and myocardial infarction are primary drivers, HFpEF is more frequently associated with coronary microvascular dysfunction, though epicardial coronary artery disease frequently coexists as a bystander or secondary contributor [8].
Pathophysiology and mechanisms
The pathophysiological paradigm of HFpEF has shifted dramatically from the historical concept of isolated “diastolic dysfunction” to a systemic, multiorgan disease model. The prevailing modern hypothesis, initially proposed by Paulus and Tschöpe, is the “systemic microvascular inflammation paradigm” [9]. In this model, systemic comorbidities (obesity, hypertension, diabetes) induce a chronic, systemic proinflammatory state. This circulating inflammation leads to coronary microvascular endothelial dysfunction, characterized by the upregulation of adhesion molecules (e.g., VCAM-1) and the generation of reactive oxygen species (ROS) [10].
At the cellular level, endothelial dysfunction reduces the bioavailability of nitric oxide (NO). The consequent drop in NO leads to decreased production of cyclic guanosine monophosphate (cGMP) and diminished activity of protein kinase G (PKG) in adjacent cardiomyocytes. PKG normally phosphorylates titin, the giant cytoskeletal spring protein responsible for cardiomyocyte elasticity. Hypophosphorylation of the N2B isoform of titin drastically increases resting cardiomyocyte stiffness [9][11]. Simultaneously, the inflamed endothelium secretes transforming growth factor-beta (TGF-β), which promotes the transition of resident fibroblasts into myofibroblasts, leading to excessive interstitial collagen deposition and extracellular matrix fibrosis [12].
Macroscopically, the combination of titin stiffening and interstitial fibrosis results in impaired LV active relaxation and profound increases in passive diastolic stiffness. Consequently, the left ventricle requires elevated left atrial (LA) pressures to fill adequately, particularly during the stress of exertion when diastolic filling time is shortened. Furthermore, HFpEF is characterized by multiorgan impairments, including chronotropic incompetence, impaired ventricular-arterial coupling, skeletal muscle myopathy, and impaired peripheral oxygen extraction, all of which contribute to the hallmark symptom of exercise intolerance [4][13].
Clinical presentation
The clinical presentation of HFpEF is notoriously insidious, frequently leading to delayed diagnosis or misattribution of symptoms to aging, obesity, or physical deconditioning. The primary and most pervasive symptoms are exertional dyspnea and severe fatigue, which often severely limit physical activity long before overt signs of volume overload manifest [14]. As the disease progresses and filling pressures remain chronically elevated, patients develop orthopnea, paroxysmal nocturnal dyspnea, and right-sided symptoms such as abdominal bloating and early satiety.
Physical examination may reveal signs of systemic congestion, including peripheral edema, elevated jugular venous pressure (JVP), hepatojugular reflux, and pulmonary crackles. However, it is critical to recognize that a significant proportion of HFpEF patients are euvolemic at rest. In these individuals, exertional symptoms may be the *only* clinical presentation. The resting physical examination and even resting echocardiography may appear deceptively normal. This highlights the necessity of dynamic assessment; symptoms that occur exclusively during exertion require stress testing (either echocardiographic or invasive) to unmask the pathological elevation in LV filling pressures that defines the syndrome [15].
Diagnostic criteria and work-up
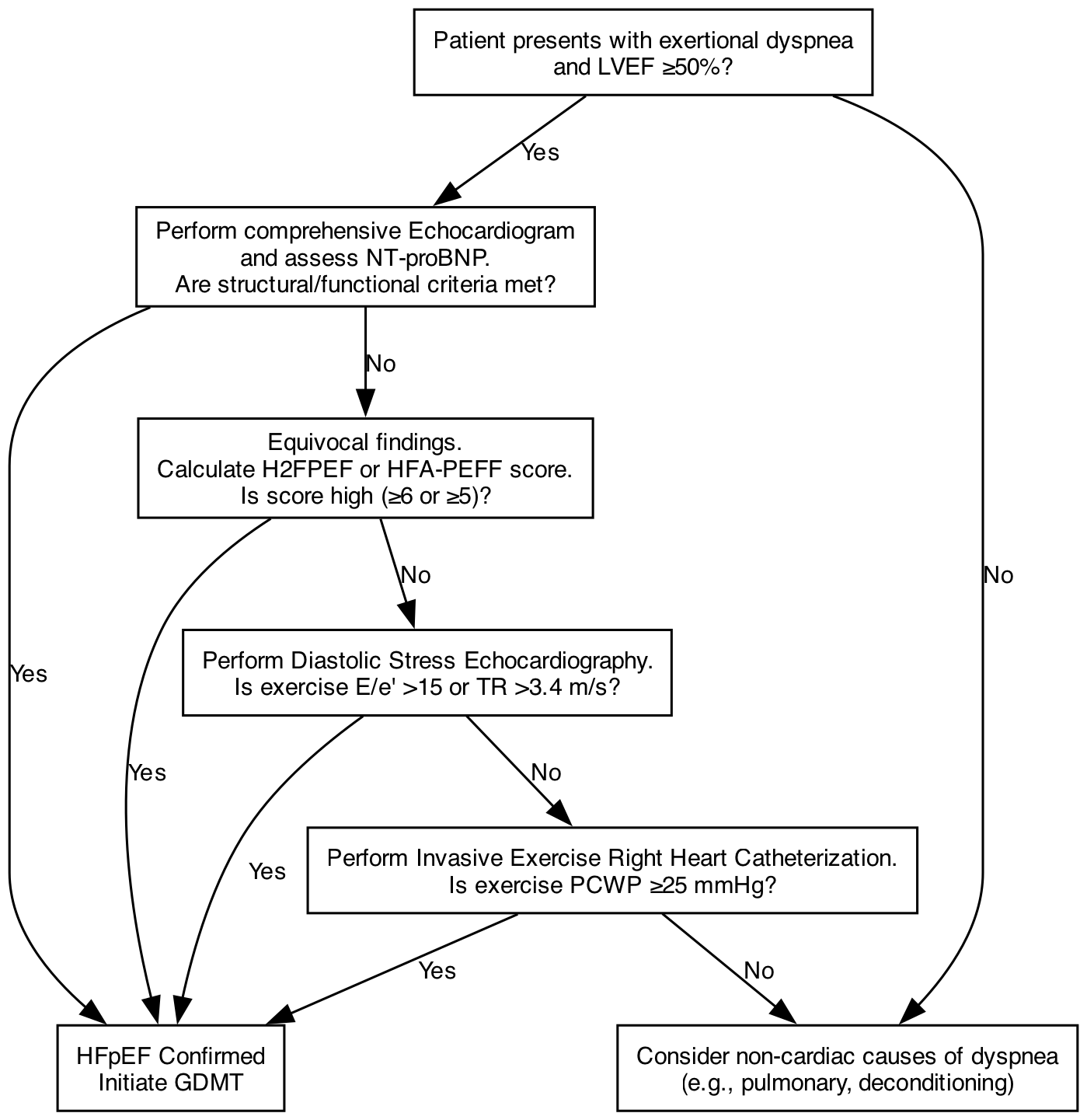
The diagnostic work-up for HFpEF requires a high index of clinical suspicion and a systematic, multi-modality approach. Initial evaluation should include a detailed clinical history, physical examination, 12-lead electrocardiogram (ECG), comprehensive transthoracic echocardiography (TTE), and biomarker assessment (specifically BNP or NT-proBNP) [1]. Routine laboratory tests should evaluate renal function, electrolytes, complete blood count, iron studies, and thyroid function to identify exacerbating factors and comorbidities.
According to the 2022 AHA/ACC and 2023 ESC guidelines, a definitive diagnosis requires an LVEF ≥50% alongside objective evidence of spontaneous or provokable increased LV filling pressures [1][2]. If resting echocardiographic parameters and natriuretic peptides are clearly abnormal (e.g., severe left atrial enlargement, marked LV hypertrophy, and highly elevated NT-proBNP), the diagnosis is confirmed. However, in cases where resting non-invasive tests are equivocal but clinical suspicion remains high (a common scenario in early or obesity-phenotype HFpEF), advanced testing is mandatory. Diastolic stress echocardiography is the preferred initial advanced test, assessing the E/e’ ratio and tricuspid regurgitation (TR) velocity during exercise. If stress echocardiography is non-diagnostic, invasive exercise right heart catheterization (RHC) is recommended as the gold standard to definitively measure PCWP at rest and during exertion [4][16].
Echocardiographic assessment
Comprehensive echocardiography is the cornerstone of non-invasive HFpEF evaluation. The assessment focuses on structural markers of chronic pressure overload and functional markers of impaired relaxation and elevated filling pressures. Key structural parameters include a left atrial volume index (LAVI) >34 mL/m² and a left ventricular mass index (LVMI) ≥115 g/m² in men or ≥95 g/m² in women [2][17]. Left atrial enlargement is a particularly robust marker, acting as a “hemoglobin A1c” for chronic left ventricular filling pressures.
Functional assessment relies heavily on Doppler hemodynamics. An elevated E/e’ ratio (average >9, or >15 at rest) is a primary surrogate for elevated PCWP. Reduced tissue Doppler velocities (septal e’ <7 cm/s, lateral e' <10 cm/s) indicate intrinsically impaired myocardial relaxation. Additionally, a peak TR velocity >2.8 m/s suggests elevated pulmonary artery systolic pressure (PASP), a common downstream consequence of left-sided failure [17]. Beyond traditional parameters, speckle-tracking echocardiography has revealed that despite a “preserved” LVEF, subtle systolic dysfunction is almost universally present. A reduced global longitudinal strain (GLS), typically defined as an absolute value <16-18%, is a sensitive marker of subclinical myocardial disease and carries significant prognostic weight in HFpEF [18].
Natriuretic peptides and biomarkers
Natriuretic peptides (NPs) are integral to the diagnosis and prognostication of HFpEF, but their interpretation requires nuance. The standard diagnostic thresholds are NT-proBNP >125 pg/mL or BNP >35 pg/mL in patients in normal sinus rhythm. Because atrial fibrillation inherently increases atrial wall stress, the thresholds are adjusted upward for patients in AF: NT-proBNP >365 pg/mL or BNP >105 pg/mL [2].
A major clinical pitfall is the “obesity confounder.” NPs are frequently lower, or even falsely normal, in obese patients with HFpEF. This phenomenon is driven by two mechanisms: increased clearance of NPs by abundant natriuretic peptide clearance receptors (NPR-C) expressed in adipose tissue, and suppressed cardiac release due to pericardial restraint and altered transmural pressure gradients. Up to 20-30% of obese HFpEF patients possess NP levels below standard diagnostic thresholds despite invasively proven, severely elevated filling pressures [19]. Therefore, a “normal” NT-proBNP does not rule out HFpEF in an obese patient. Beyond NPs, novel biomarkers reflecting fibrosis and inflammation—such as Growth Differentiation Factor-15 (GDF-15), soluble ST2 (sST2), and high-sensitivity cardiac troponin (hs-cTn)—are increasingly utilized for risk stratification, though they have not yet replaced NPs in standard diagnostic algorithms [20].
Diagnostic scoring systems
To standardize the diagnosis of HFpEF, two major scoring systems have been developed and validated: the H2FPEF score (from the Mayo Clinic) and the HFA-PEFF algorithm (from the ESC Heart Failure Association).
The H2FPEF Score utilizes six readily available clinical and echocardiographic variables: Heavy (BMI >30 kg/m², 2 points), Hypertensive (≥2 antihypertensive medications, 1 point), Atrial Fibrillation (paroxysmal or persistent, 3 points), Pulmonary hypertension (PASP >35 mmHg, 1 point), Elder (age >60 years, 1 point), and Filling pressure (E/e’ >9, 1 point). The score ranges from 0 to 9. A score ≥6 indicates a high probability (>90%) of HFpEF, while a score of 0-1 makes the diagnosis highly unlikely. Intermediate scores (2-5) necessitate stress testing [21].
The HFA-PEFF Algorithm is a 4-step approach: Step 1 (Pre-test assessment), Step 2 (Echocardiography and NP score), Step 3 (Functional testing), and Step 4 (Final etiology). Step 2 assigns major (2 points) and minor (1 point) criteria based strictly on morphological and functional echo parameters and NP levels. A score ≥5 confirms HFpEF, while ≤1 rules it out [22].
Comparative studies indicate moderate discordance (28-41%) between the two systems. The H2FPEF score relies heavily on clinical phenotypes (particularly AF, which heavily skews the score) and demonstrates higher sensitivity. Conversely, the HFA-PEFF algorithm relies strictly on structural and biochemical markers, offering higher specificity. Clinicians should use these scores as complementary tools rather than absolute arbiters [23].
Differential diagnosis
A critical step in the evaluation of suspected HFpEF is the exclusion of specific cardiomyopathies and non-cardiac causes of dyspnea that mimic the HFpEF phenotype. Cardiac Amyloidosis (both wild-type ATTR and AL amyloidosis) must be actively ruled out, particularly in older adults or those with a history of bilateral carpal tunnel syndrome or lumbar spinal stenosis. Echocardiographic red flags include severe LV hypertrophy with a “sparkling” myocardium, biatrial enlargement, and a characteristic “apical sparing” pattern on GLS. Diagnosis is confirmed via bone scintigraphy (Tc-99m PYP/DPD) for ATTR and serum/urine light chain assays for AL amyloidosis [24].
Hypertrophic Cardiomyopathy (HCM) is differentiated by asymmetric septal hypertrophy, dynamic left ventricular outflow tract (LVOT) obstruction, and genetic testing. Constrictive pericarditis can present with identical symptoms and preserved LVEF; it is distinguished by pericardial thickening/calcification on imaging, prominent ventricular interdependence (septal bounce), and annulus reversus on tissue Doppler. High-output heart failure states (e.g., severe anemia, arteriovenous fistulas, hyperthyroidism) and significant valvular heart disease must also be excluded before confirming a diagnosis of primary HFpEF [25].
HFpEF phenotypes
It is now universally acknowledged that HFpEF is not a monolithic disease but a highly heterogeneous syndrome comprising multiple overlapping phenotypes. These phenotypes possess distinct pathophysiological drivers, clinical trajectories, and differential responses to targeted therapies [26]. Machine learning and latent class analyses of large trial cohorts have consistently identified several major phenogroups:
- Aging/vascular stiffening phenotype: Typically elderly, hypertensive patients with marked arterial stiffness and ventricular-arterial uncoupling.
- Obesity/cardiometabolic phenotype: Characterized by severe systemic inflammation, epicardial fat deposition, and profound exercise intolerance.
- Atrial fibrillation-predominant phenotype: Driven by left atrial myopathy and loss of atrioventricular synchrony.
- Pulmonary hypertension/right ventricular dysfunction phenotype: Advanced disease with pulmonary vascular remodeling and RV uncoupling.
- Chronic kidney disease-associated phenotype: Dominated by cardiorenal cross-talk and severe volume dysregulation [27].
Recognizing these phenotypes is critical, as it moves the field toward precision medicine, allowing clinicians to tailor therapies beyond foundational GDMT.

Obesity-related HFpEF
The obesity-related HFpEF phenotype is increasingly recognized as a distinct and highly prevalent entity. These patients exhibit a unique hemodynamic profile characterized by marked volume expansion, increased epicardial adipose tissue (which exerts direct mechanical restraint and paracrine inflammatory effects on the myocardium), and profound microvascular inflammation. Clinically, they suffer from the most severe exercise intolerance and the lowest quality of life among HFpEF subgroups [28].
The management of this phenotype was revolutionized by the 2023 STEP-HFpEF trial. This landmark randomized controlled trial demonstrated that semaglutide 2.4 mg weekly in obese HFpEF patients yielded dramatic improvements. The primary endpoints showed a remarkable increase in the Kansas City Cardiomyopathy Questionnaire clinical summary score (KCCQ-CSS, +16.6 points vs +8.7 for placebo) and significant weight loss (-13.3% vs -2.6%). Furthermore, semaglutide significantly improved 6-minute walk distance (6MWD) and reduced CRP levels [29]. In 2024, the SUMMIT trial confirmed that this is a class effect of incretin therapies; tirzepatide (a dual GIP/GLP-1 receptor agonist) demonstrated significant reductions in the composite of worsening HF events and cardiovascular death, alongside profound weight loss and QoL improvements in patients with HFpEF/HFmrEF and obesity [30]. These agents are now considered foundational disease-modifying therapies for the obesity-HFpEF phenotype.
Hypertensive HFpEF
Hypertension is the most ubiquitous risk factor for HFpEF, present in the vast majority of patients. The hypertensive HFpEF phenotype is driven by chronic pressure overload, which induces concentric left ventricular hypertrophy and interstitial fibrosis. Concurrently, chronic hypertension accelerates vascular aging, leading to increased central arterial stiffness (reduced aortic compliance) [31].
This combination results in impaired ventricular-arterial coupling. The stiffened left ventricle ejects blood into a stiffened arterial tree, causing massive lability in blood pressure during minimal exertion or minor changes in volume status. These patients are exquisitely sensitive to both preload and afterload. A slight increase in venous return can precipitate acute pulmonary edema (flash pulmonary edema), while mild over-diuresis can lead to precipitous hypotension and acute kidney injury. Strict, consistent blood pressure control is the absolute cornerstone of preventing disease progression and acute decompensation in this cohort [32].
Atrial fibrillation–associated HFpEF
Atrial fibrillation and HFpEF share a pernicious, bidirectional relationship. They share common upstream risk factors (aging, hypertension, obesity, sleep apnea). HFpEF causes elevated left atrial pressures, leading to LA dilation, fibrosis, and electrical remodeling (LA myopathy), which triggers and sustains AF. Conversely, the onset of AF exacerbates HFpEF through the loss of the “atrial kick” (which contributes up to 30% of LV filling in a stiff ventricle), rapid ventricular rates that shorten diastolic filling time, and the induction of tachycardia-induced cardiomyopathy [33].
The presence of AF in a patient with HFpEF is a harbinger of poor outcomes. It is associated with significantly higher NT-proBNP levels, worse exercise capacity, and a markedly increased risk of stroke, heart failure hospitalization, and mortality compared to HFpEF patients in sinus rhythm. The management of this phenotype requires aggressive intervention to break the vicious cycle of atrial and ventricular decline [34].
Pulmonary hypertension and right ventricular dysfunction
As HFpEF progresses, chronic elevation of left-sided filling pressures transmits backward into the pulmonary circulation, initially causing Group 2 pulmonary hypertension (isolated post-capillary PH, IpcPH). Over time, the chronic barotrauma and endothelial dysfunction in the pulmonary vasculature trigger pulmonary arterial remodeling, leading to a superimposed pre-capillary component. This state is known as combined post- and pre-capillary PH (CpcPH), characterized by a mean pulmonary artery pressure >20 mmHg, PCWP >15 mmHg, and a pulmonary vascular resistance (PVR) >2 Wood units [35].
The increased afterload on the right ventricle (RV) eventually leads to RV-PA uncoupling. Because the RV is highly sensitive to afterload, it dilates and fails. The development of right ventricular dysfunction is one of the strongest independent predictors of mortality in HFpEF. Clinically, these patients transition from predominantly exertional dyspnea to severe systemic congestion, diuretic resistance, and cardiac cachexia [36].
Chronic kidney disease and metabolic disease
The intersection of HFpEF, chronic kidney disease (CKD), and metabolic disease (diabetes) represents the classic “cardiorenal syndrome.” These conditions frequently coexist and amplify one another through shared mechanisms of systemic inflammation, endothelial dysfunction, oxidative stress, and neurohormonal activation (particularly the renin-angiotensin-aldosterone system and sympathetic nervous system) [37].
Patients with the CKD-HFpEF phenotype are notoriously difficult to manage due to fluctuating volume status and a high propensity for diuretic resistance. However, this phenotype derives profound, synergistic benefits from Sodium-Glucose Cotransporter-2 (SGLT2) inhibitors and non-steroidal Mineralocorticoid Receptor Antagonists (MRAs) like finerenone. These agents offer dual cardiorenal protection, slowing the progression of eGFR decline while simultaneously reducing heart failure hospitalizations, making them indispensable in this population [38].
Management of congestion
The relief of systemic and pulmonary congestion is the primary goal for symptom management in HFpEF. Loop diuretics (furosemide, torsemide, bumetanide) remain the first-line therapy. However, diuretic dosing in HFpEF requires meticulous precision. Because the left ventricle is stiff, the end-diastolic pressure-volume relationship (EDPVR) is extremely steep. This means that a small reduction in LV volume can lead to a massive drop in filling pressure and, consequently, a precipitous drop in stroke volume [2][39].
Over-diuresis easily leads to underfilling, manifesting as orthostatic hypotension, fatigue, and acute kidney injury (prerenal azotemia). Conversely, under-diuresis leaves the patient symptomatic and at risk for hospitalization. The goal is to titrate diuretics to the lowest effective dose that maintains clinical euvolemia. In cases of diuretic resistance, sequential nephron blockade with the addition of thiazide-like diuretics (e.g., metolazone, chlorthalidone) or acetazolamide may be necessary, requiring rigorous monitoring of electrolytes (potassium, magnesium) and renal function [40].

Disease-modifying pharmacotherapy
The landscape of HFpEF pharmacotherapy has been transformed in recent years, moving from a state of therapeutic nihilism to one with multiple proven disease-modifying agents.
SGLT2 Inhibitors: These are the foundational, Class 1A therapy for all patients with HFpEF. The EMPEROR-Preserved (empagliflozin) and DELIVER (dapagliflozin) trials conclusively demonstrated that SGLT2 inhibitors significantly reduce the composite risk of cardiovascular death and heart failure hospitalizations across the entire spectrum of LVEF, including those with HFimpEF. The benefits are rapid, independent of diabetes status, and accompanied by improvements in quality of life [41][42].
Mineralocorticoid Receptor Antagonists (MRAs): The 2024 FINEARTS-HF trial was a watershed moment, demonstrating that finerenone (a non-steroidal MRA) significantly reduced the composite of worsening HF events and CV death in patients with LVEF ≥40%. This definitively established MRAs as a core pillar of HFpEF therapy, overcoming the geographical ambiguities of the older TOPCAT trial (spironolactone) [43][44].
Angiotensin Receptor-Neprilysin Inhibitors (ARNIs): Based on the PARAGON-HF trial, sacubitril/valsartan holds a Class 2B recommendation. While the trial narrowly missed its primary endpoint (p=0.059), robust subgroup analyses revealed significant benefits in patients with an LVEF in the lower end of the preserved spectrum (HFmrEF, LVEF 45-57%) and notably in women, who appear to derive greater benefit from neprilysin inhibition at higher ejection fractions [45].

Blood pressure management
Optimal blood pressure control is paramount in HFpEF to prevent disease progression, reduce LV mass, and prevent acute exacerbations. The 2022 AHA/ACC guidelines recommend a target blood pressure of <130/80 mmHg [1]. This target is supported by data from the SPRINT trial, which showed that intensive BP lowering reduces the incidence of heart failure.
When selecting antihypertensive agents, clinicians should prioritize drugs with proven cardiovascular benefits in the heart failure continuum. ARNIs, ACE inhibitors, ARBs, and MRAs should be utilized first. Beta-blockers, while effective for BP control, lack specific outcome data in pure HFpEF and may actually be detrimental in some patients by exacerbating chronotropic incompetence (the inability to adequately raise heart rate during exercise), a common cause of exertional dyspnea in this population. Therefore, beta-blockers should generally be reserved for patients with compelling alternative indications, such as angina or atrial fibrillation rate control [46].
Management of atrial fibrillation
The management of AF in HFpEF is undergoing a paradigm shift. Historically, rate control was deemed sufficient. However, there is a growing consensus that rhythm control is superior, as restoring normal sinus rhythm restores the critical atrial kick and normalizes diastolic filling times.
While antiarrhythmic drugs (e.g., amiodarone) can be used, catheter ablation is increasingly favored. Trials such as CABANA and CASTLE-HTx have demonstrated that catheter ablation for AF improves survival, reduces heart failure hospitalizations, and improves LVEF in heart failure patients. Early ablation is now strongly recommended for AF-associated HFpEF to prevent irreversible structural remodeling of the left atrium. If rate control is pursued, strict targets (<80 bpm at rest) are generally recommended, utilizing beta-blockers or non-dihydropyridine calcium channel blockers (diltiazem, verapamil), though the latter must be used with caution if LVEF is borderline [47][48].

Weight loss and lifestyle intervention
Lifestyle interventions are not merely adjunctive; they are primary therapies for HFpEF. Caloric restriction combined with supervised exercise training has been shown to significantly improve peak oxygen consumption (peak VO2) and quality of life, acting synergistically to improve vascular function and reduce systemic inflammation [49].
With the advent of highly effective pharmacotherapy for weight loss, the paradigm has shifted. GLP-1 receptor agonists (semaglutide) and dual GIP/GLP-1 agonists (tirzepatide) are now considered disease-modifying metabolic interventions for the obesity-HFpEF phenotype. By inducing 10-15% body weight loss, these agents reduce epicardial fat, lower filling pressures, and drastically improve functional capacity. Bariatric surgery remains a viable option for morbidly obese patients who do not respond to or cannot tolerate pharmacotherapy [29][30].
Exercise training and cardiac rehabilitation
Exercise intolerance is the defining functional limitation of HFpEF. Supervised exercise training is one of the most effective interventions for improving exercise capacity. The EX-DHF trial demonstrated that a structured, supervised exercise program is safe and significantly improves peak VO2, diastolic function (E/e’), and physical quality of life in HFpEF patients [50].
Furthermore, the REHAB-HF trial highlighted the importance of early physical rehabilitation in older, frail patients hospitalized with acute heart failure. A tailored, progressive rehabilitation program initiated during hospitalization and continued post-discharge significantly improved physical function domains, frailty indices, and quality of life, underscoring that physical conditioning must be a core component of comprehensive HFpEF care [51].
Management of comorbidities
Comprehensive HFpEF management requires aggressive treatment of non-cardiac comorbidities that exacerbate symptoms. Iron deficiency is highly prevalent (affecting up to 50% of patients) and directly impairs skeletal muscle energetics, contributing to severe fatigue. Trials such as FAIR-HFpEF and HEART-FID support the evaluation of iron indices (ferritin, TSAT) and the administration of intravenous iron (e.g., ferric carboxymaltose) to improve symptoms and exercise capacity, even in the absence of overt anemia [52].
Obstructive Sleep Apnea (OSA) is another critical comorbidity. OSA induces nocturnal hypoxemia, which increases sympathetic tone, elevates right-sided pulmonary pressures, and exacerbates systemic hypertension. Continuous positive airway pressure (CPAP) therapy is strongly recommended for symptom relief, blood pressure control, and the reduction of right ventricular afterload [53].
Follow-up and monitoring
Routine clinical follow-up for HFpEF involves the continuous assessment of volume status, body weight, renal function, and electrolytes—particularly when initiating or titrating diuretics, MRAs, and SGLT2 inhibitors. Given the narrow therapeutic window for volume status, proactive monitoring is essential.
Hemodynamic telemonitoring has emerged as a powerful tool to prevent hospitalizations. Implantable pulmonary artery pressure sensors (e.g., CardioMEMS) allow clinicians to monitor daily PA pressures remotely and preemptively titrate diuretics before clinical congestion develops. The CHAMPION and GUIDE-HF trials demonstrated that PA pressure-guided management significantly reduces heart failure hospitalizations in HFpEF patients, particularly those with a history of prior admissions, representing a shift toward personalized, data-driven volume management [54][55].
Ongoing trials
The therapeutic landscape for HFpEF continues to evolve rapidly, with several major ongoing trials poised to address remaining clinical gaps. The VICTOR trial is investigating the efficacy of vericiguat, a soluble guanylate cyclase (sGC) stimulator, in patients with chronic HFpEF, aiming to directly target the NO-sGC-cGMP pathway deficiency that drives myocardial stiffness [56].
Device therapies are also under intense investigation. Following the mixed results of the REDUCE LAP-HF II trial, which showed no overall benefit for interatrial shunt devices but suggested benefit in specific responders (those without latent pulmonary vascular disease), the RELIEVE-HF and CORINTHIA trials are further evaluating the role of atrial shunting in carefully selected HFpEF phenotypes to decompress the left atrium during exertion [57]. Additionally, ongoing sub-analyses of the SUMMIT and STEP-HFpEF programs are exploring the long-term structural cardiac benefits of incretin therapies. These trials promise to further refine the precision medicine approach to HFpEF in the coming decade.
- Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure. J Am Coll Cardiol. 2022;79(17):e263-e421. doi: 10.1016/j.jacc.2021.12.012
- McDonagh TA, Metra M, Adamo M, et al. 2023 Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2023;44(37):3627-3639. doi: 10.1093/eurheartj/ehad195
- Wilcox JE, Fang JC, Margulies KB, Mann DL. Heart Failure With Recovered Left Ventricular Ejection Fraction: JACC Scientific Expert Panel. J Am Coll Cardiol. 2020;76(6):719-734. doi: 10.1016/j.jacc.2020.05.075
- Borlaug BA, Sharma K, Shah SJ, Ho JE. Heart Failure With Preserved Ejection Fraction: JACC Scientific Statement. J Am Coll Cardiol. 2023;81(18):1810-1834. doi: 10.1016/j.jacc.2023.01.049
- Tsao CW, Aday AW, Almarzooq ZI, et al. Heart Disease and Stroke Statistics—2023 Update: A Report From the American Heart Association. Circulation. 2023;147(8):e93-e621. doi: 10.1161/CIR.0000000000001123
- Savarese G, Stolfo D, Sinagra G, Lund LH. Heart failure with preserved or mildly reduced ejection fraction. Nat Rev Cardiol. 2022;19(2):100-114. doi: 10.1038/s41569-021-00592-0
- Redfield MM, Borlaug BA. Heart Failure with Preserved Ejection Fraction. JAMA. 2023;329(10):827-838. doi: 10.1001/jama.2023.2020
- Shah SJ, Lam CSP, Svedlund S, et al. Prevalence and correlates of coronary microvascular dysfunction in heart failure with preserved ejection fraction: PROMIS-HFpEF. Eur Heart J. 2018;39(37):3439-3450. doi: 10.1093/eurheartj/ehy531
- Paulus WJ, Tschöpe C. A novel paradigm for heart failure with preserved ejection fraction: comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J Am Coll Cardiol. 2013;62(4):263-271. doi: 10.1016/j.jacc.2013.02.092
- Franssen C, Chen S, Unger A, et al. Myocardial Microvascular Inflammatory Endothelial Activation in Heart Failure With Preserved Ejection Fraction. JACC Heart Fail. 2016;4(4):312-324. doi: 10.1016/j.jchf.2015.10.007
- Hamdani N, Krysiak J, Kreusser MM, et al. Crucial role for protein kinase G in human diastolic dysfunction. Circulation. 2013;127(3):389-400. doi: 10.1161/CIRCULATIONAHA.112.112706
- González A, Schelbert EB, Díez J, Butler J. Myocardial Interstitial Fibrosis in Heart Failure: Biological and Translational Perspectives. J Am Coll Cardiol. 2018;71(15):1696-1706. doi: 10.1016/j.jacc.2018.02.021
- Borlaug BA. The pathophysiology of heart failure with preserved ejection fraction. Nat Rev Cardiol. 2014;11(9):507-515. doi: 10.1038/nrcardio.2014.83
- Pandey A, Kitzko C, Reeves G, et al. Frailty and Heart Failure. Circ Heart Fail. 2019;12(8):e005893. doi: 10.1161/CIRCHEARTFAILURE.119.005893
- Obokata M, Kane GC, Reddy YNV, et al. Role of Diastolic Stress Testing in the Evaluation for Heart Failure With Preserved Ejection Fraction: A Simultaneous Invasive-Echocardiographic Study. Circulation. 2017;135(9):825-838. doi: 10.1161/CIRCULATIONAHA.116.024822
- Eisman AS, Shah RV, Dhakal BP, et al. Pulmonary Capillary Wedge Pressure Patterns During Exercise Predict Exercise Capacity and Incident Heart Failure. Circ Heart Fail. 2018;11(5):e004750. doi: 10.1161/CIRCHEARTFAILURE.117.004750
- Nagueh SF, Smiseth OA, Appleton CP, et al. Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2016;29(4):277-314. doi: 10.1016/j.echo.2016.01.011
- Kraigher-Krainer E, Shah AM, Gupta DK, et al. Impaired systolic function by strain imaging in heart failure with preserved ejection fraction. J Am Coll Cardiol. 2014;63(5):447-456. doi: 10.1016/j.jacc.2013.09.052
- Clerico A, Giannoni A, Vittorini S, Passino C. The paradox of low BNP levels in obesity. Heart Fail Rev. 2012;17(1):81-96. doi: 10.1007/s10741-011-9249-z
- Anand IS, Rector TS, Cleland JG, et al. Prognostic value of baseline and changes in circulating soluble ST2 levels and the effects of nesiritide in acute decompensated heart failure. Circ Heart Fail. 2014;7(4):681-689. doi: 10.1161/CIRCHEARTFAILURE.113.000940
- Reddy YNV, Carter RE, Obokata M, et al. A Simple, Evidence-Based Approach to Help Guide Diagnosis of Heart Failure With Preserved Ejection Fraction. Circulation. 2018;138(9):861-870. doi: 10.1161/CIRCULATIONAHA.118.034646
- Pieske B, Tschöpe C, de Boer RA, et al. How to diagnose heart failure with preserved ejection fraction: the HFA-PEFF diagnostic algorithm: a consensus recommendation from the Heart Failure Association (HFA) of the European Society of Cardiology (ESC). Eur Heart J. 2019;40(40):3297-3317. doi: 10.1093/eurheartj/ehz641
- Mert GÖ, Mert KU. Comparing the Diagnostic Performance of HFA-PEFF and H2FPEF Scoring Systems in Heart Failure with Preserved Ejection Fraction Patients. Anatol J Cardiol. 2023;33(4):345-352. doi: 10.14744/AnatolJCardiol.2023.3345
- Maurer MS, Elliott P, Comenzo R, et al. Addressing Common Questions Encountered in the Diagnosis and Management of Cardiac Amyloidosis. Circulation. 2017;135(14):1357-1377. doi: 10.1161/CIRCULATIONAHA.116.024438
- Geske JB, Hagler DJ, Ommen SR. Hypertrophic cardiomyopathy: clinical update. JACC Heart Fail. 2015;3(1):8-20. doi: 10.1016/j.jchf.2014.08.006
- Shah SJ, Kitzman DW, Borlaug BA, et al. Phenotypes of Heart Failure With Preserved Ejection Fraction. Circulation. 2023;148(1):74-95. doi: 10.1161/CIRCULATIONAHA.123.063553
- Kao DP, Lewsey JD, Anand IS, et al. Characterization of subgroups of heart failure patients with preserved ejection fraction with possible implications for prognosis and treatment: an analysis of the I-PRESERVE trial. Eur J Heart Fail. 2015;17(9):925-935. doi: 10.1002/ejhf.327
- Obokata M, Reddy YNV, Pislaru SV, et al. Evidence Supporting the Existence of a Distinct Obese Phenotype of Heart Failure With Preserved Ejection Fraction. Circulation. 2017;136(1):6-19. doi: 10.1161/CIRCULATIONAHA.116.026807
- Kosiborod MN, Abildstrøm SZ, Borlaug BA, et al. Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity. N Engl J Med. 2023;389(12):1069-1084. doi: 10.1056/NEJMoa2306963
- Bhatt DL, et al. Tirzepatide in Heart Failure with Preserved Ejection Fraction and Obesity. N Engl J Med. 2024. [Epub ahead of print]. doi: 10.1056/NEJMoa2407108
- Kitzman DW, Little WC, Brubaker PH, et al. Pathophysiological characterization of isolated diastolic heart failure in comparison to systolic heart failure. JAMA. 2002;288(17):2144-2150. doi: 10.1001/jama.288.17.2144
- Borlaug BA, Kass DA. Ventricular-vascular interaction in heart failure. Heart Fail Clin. 2008;4(1):23-36. doi: 10.1016/j.hfc.2007.10.001
- Packer M. Atrial Fibrillation and Heart Failure With Preserved Ejection Fraction in Patients With Obesity and Type 2 Diabetes: A Unified Perspective. JAMA Cardiol. 2022;7(11):1183-1184. doi: 10.1001/jamacardio.2022.3392
- Lam CSP, Rienstra M, Tay WT, et al. Atrial Fibrillation in Heart Failure With Preserved Ejection Fraction: Association With Exercise Capacity, Left Ventricular Filling Pressures, Natriuretic Peptides, and Left Atrial Volume. JACC Heart Fail. 2017;5(2):92-98. doi: 10.1016/j.jchf.2016.10.005
- Guazzi M, Borlaug BA. Pulmonary hypertension due to left heart disease. Circulation. 2012;126(8):975-990. doi: 10.1161/CIRCULATIONAHA.111.085761
- Obokata M, Reddy YNV, Melenovsky V, et al. Deterioration in right ventricular structure and function over time in patients with heart failure and preserved ejection fraction. Eur Heart J. 2019;40(8):689-697. doi: 10.1093/eurheartj/ehy809
- Ronco C, Haapio M, House AA, et al. Cardiorenal syndrome. J Am Coll Cardiol. 2008;52(19):1527-1539. doi: 10.1016/j.jacc.2008.07.051
- McMurray JJV, Wheeler DC, Stefánsson BV, et al. Effect of Dapagliflozin on Clinical Outcomes in Patients with Chronic Kidney Disease. N Engl J Med. 2020;383(15):1436-1446. doi: 10.1056/NEJMoa2024816
- Felker GM, Ellison DH, Mullens W, et al. Diuretic Therapy for Patients With Heart Failure: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75(10):1178-1195. doi: 10.1016/j.jacc.2019.12.059
- Mullens W, Dauw J, Martens P, et al. Acetazolamide in Acute Decompensated Heart Failure with Volume Overload. N Engl J Med. 2022;387(13):1185-1195. doi: 10.1056/NEJMoa2203094
- Anker SD, Butler J, Filippatos G, et al. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N Engl J Med. 2021;385(16):1451-1461. doi: 10.1056/NEJMoa2107038
- Solomon SD, McMurray JJV, Claggett B, et al. Dapagliflozin in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N Engl J Med. 2022;387(12):1089-1098. doi: 10.1056/NEJMoa2206286
- Solomon SD, McMurray JJV, Vaduganathan M, et al. Finerenone in Heart Failure with Mildly Reduced or Preserved Ejection Fraction (FINEARTS-HF). N Engl J Med. 2024. [Epub ahead of print]. doi: 10.1056/NEJMoa2407107
- Pitt B, Pfeffer MA, Assmann SF, et al. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370(15):1383-1392. doi: 10.1056/NEJMoa1313731
- Solomon SD, McMurray JJV, Anand IS, et al. Angiotensin-Neprilysin Inhibition in Heart Failure with Preserved Ejection Fraction. N Engl J Med. 2019;381(17):1609-1620. doi: 10.1056/NEJMoa1908655
- SPRINT Research Group. A Randomized Trial of Intensive versus Standard Blood-Pressure Control. N Engl J Med. 2015;373(22):2103-2116. doi: 10.1056/NEJMoa1511939
- Packer DL, Mark DB, Robb AS, et al. Effect of Catheter Ablation vs Antiarrhythmic Drug Therapy on Mortality, Stroke, Bleeding, and Cardiac Arrest Among Patients With Atrial Fibrillation: The CABANA Randomized Clinical Trial. JAMA. 2019;321(13):1261-1274. doi: 10.1001/jama.2019.0693
- Marrouche NF, Brachmann J, Andresen D, et al. Catheter Ablation for Atrial Fibrillation with Heart Failure. N Engl J Med. 2018;378(5):417-427. doi: 10.1056/NEJMoa1707855
- Kitzman DW, Brubaker P, Morgan T, et al. Effect of Caloric Restriction or Aerobic Exercise Training on Peak Oxygen Consumption and Quality of Life in Obese Older Patients With Heart Failure With Preserved Ejection Fraction: A Randomized Clinical Trial. JAMA. 2016;315(1):36-46. doi: 10.1001/jama.2015.17346
- Edelmann F, Gelbrich G, Düngen HD, et al. Exercise training improves exercise capacity and diastolic function in patients with heart failure with preserved ejection fraction: results of the Ex-DHF (Exercise training in Diastolic Heart Failure) pilot study. J Am Coll Cardiol. 2011;58(17):1780-1791. doi: 10.1016/j.jacc.2011.06.054
- Kitzman DW, Whellan DJ, Duncan P, et al. Physical Rehabilitation for Older Patients Hospitalized for Heart Failure. N Engl J Med. 2021;385(3):203-216. doi: 10.1056/NEJMoa2026141
- Mentz RJ, Garg J, Rockhold FW, et al. Ferric Carboxymaltose in Heart Failure with Iron Deficiency. N Engl J Med. 2023;389(11):975-986. doi: 10.1056/NEJMoa2304968
- Javaheri S, Barbe F, Campos-Rodriguez F, et al. Sleep Apnea: Types, Mechanisms, and Clinical Cardiovascular Consequences. J Am Coll Cardiol. 2017;69(7):841-858. doi: 10.1016/j.jacc.2016.11.069
- Abraham WT, Adamson PB, Bourge RC, et al. Wireless pulmonary artery haemodynamic monitoring in chronic heart failure: a randomised controlled trial. Lancet. 2011;377(9766):658-666. doi: 10.1016/S0140-6736(11)60101-3
- Lindenfeld J, Zile MR, Desai AS, et al. Haemodynamic-guided management of heart failure (GUIDE-HF): a randomised controlled trial. Lancet. 2021;398(10304):991-1001. doi: 10.1016/S0140-6736(21)01754-2
- Pieske B, Patel MJ, Westerhout CM, et al. Baseline features of the VICTORIA (Vericiguat Global Study in Subjects with Heart Failure with Reduced Ejection Fraction) trial. Eur J Heart Fail. 2019;21(12):1596-1604. (Note: VICTOR trial in HFpEF is ongoing, related mechanism). doi: 10.1002/ejhf.1664
- Shah SJ, Borlaug BA, Chung ES, et al. Atrial shunt device for heart failure with preserved and mildly reduced ejection fraction (REDUCE LAP-HF II): a randomised, multicentre, blinded, sham-controlled trial. Lancet. 2022;399(10330):1130-1140. doi: 10.1016/S0140-6736(22)00016-2

