Atrial Fibrillation and Atrial Thromboembolism
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia and a profound driver of global cardiovascular morbidity and mortality, primarily due to its association with ischemic stroke and systemic embolism. The contemporary understanding of AF has evolved significantly; it is no longer viewed merely as an electrical anomaly but as a progressive, complex atrial myopathy. For the practicing cardiologist, mastering the nuances of anticoagulation requires an intimate understanding of the underlying pathophysiology, which is classically framed by Virchow’s triad: blood stasis, endothelial dysfunction, and hypercoagulability [1][2].
Mechanical stasis in AF is most pronounced in the left atrial appendage (LAA). The LAA is a highly trabeculated, windsock-like embryonic remnant that exhibits uniquely low flow velocities during the loss of organized atrial systole. Transesophageal echocardiography and surgical pathology demonstrate that over 90% of thrombi in non-valvular AF originate within this specific anatomical cul-de-sac [19]. Concurrently, the atrial myocardium undergoes structural remodeling driven by aging, hypertension, heart failure, and the arrhythmia itself. This remodeling manifests as interstitial fibrosis, atrial dilation, and endocardial denudation, which collectively promote local inflammation and create a highly prothrombotic endothelial surface. Furthermore, AF induces a systemic hypercoagulable state, characterized by elevated biomarkers such as D-dimer, von Willebrand factor, and increased thrombin generation.
The clinical consequence of this triad is severe: cardioembolic strokes associated with AF are typically larger, present with more severe neurological deficits, and carry higher rates of mortality and long-term disability compared to non-cardioembolic ischemic strokes. Consequently, the cornerstone of AF management remains the meticulous application of anticoagulant therapies, balancing the imperative of stroke prevention against the inherent risks of iatrogenic hemorrhage.
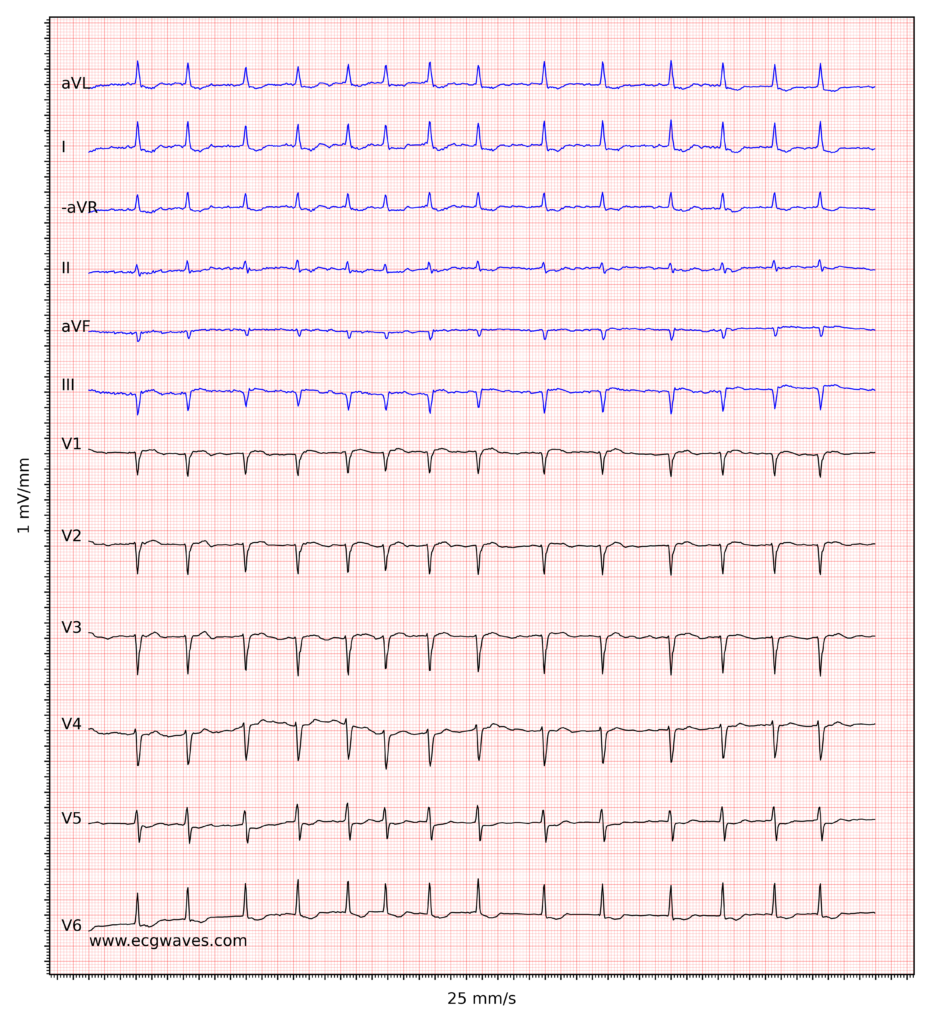
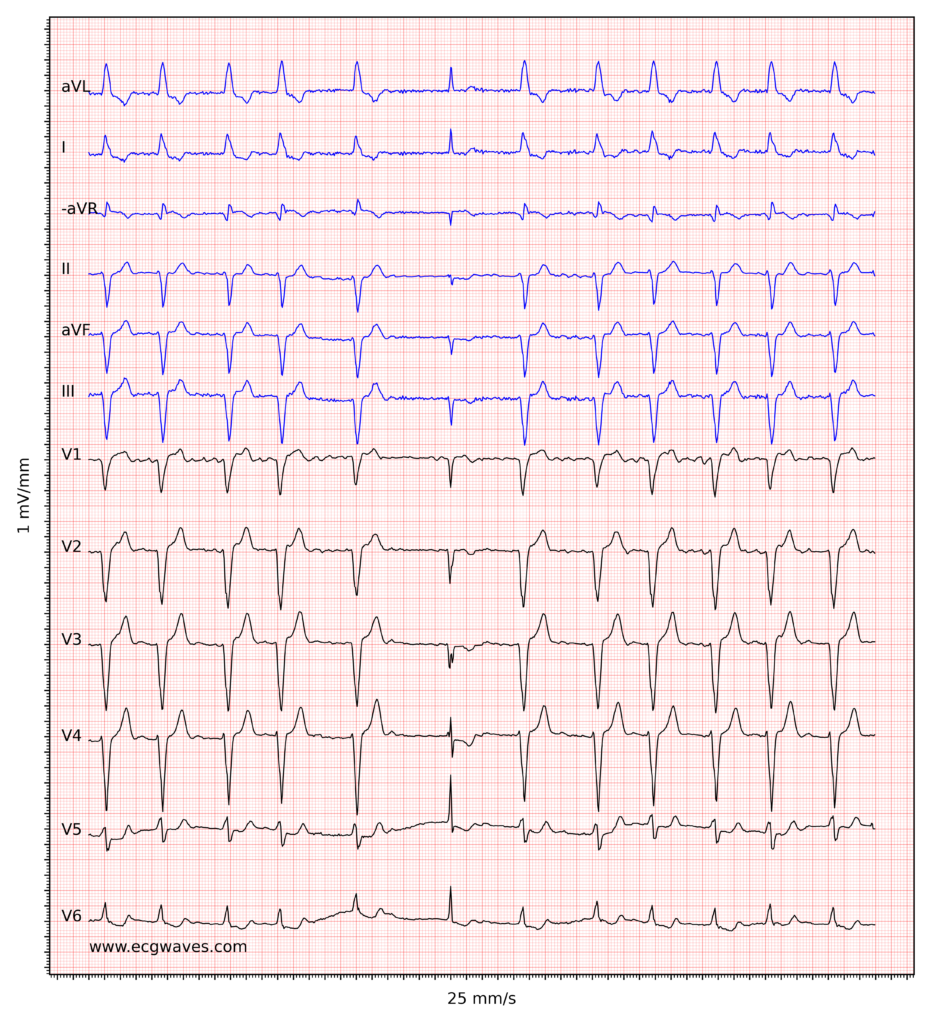
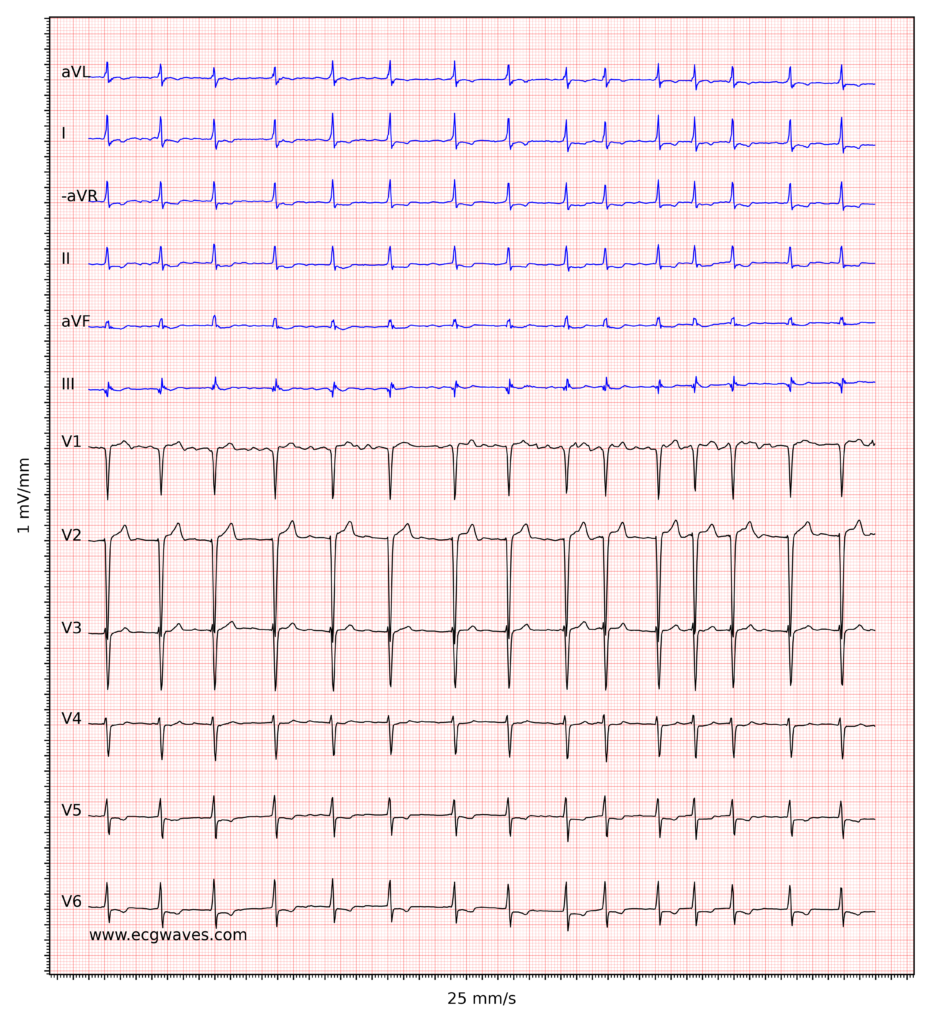
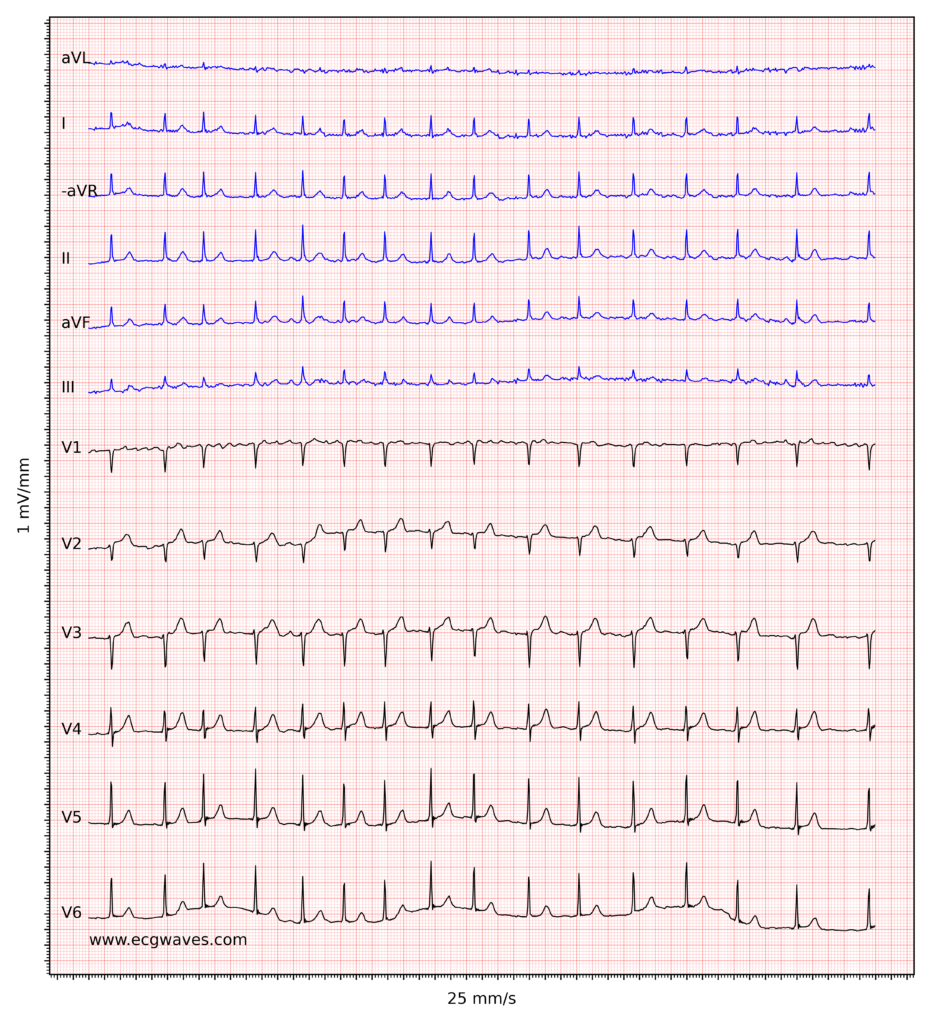
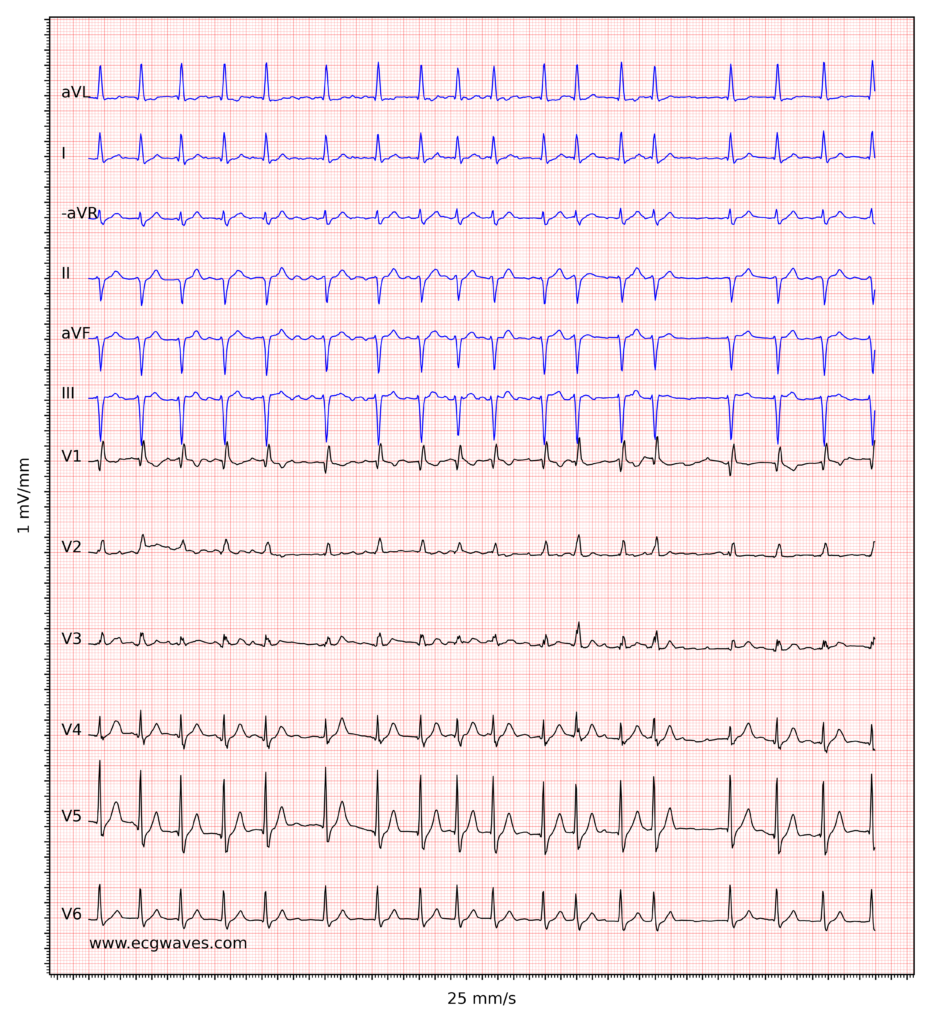
ECG recordings of atrial fibrillation. Click to enlarge.

Risk Stratification: Stroke and Bleeding Assessment
The decision to initiate oral anticoagulation (OAC) hinges on a rigorous assessment of both thromboembolic and hemorrhagic risks. The diagnostic prerequisite for clinical AF remains the documentation of the arrhythmia via a standard 12-lead ECG or a single-lead ECG tracing lasting ≥30 seconds, characterized by irregularly irregular R-R intervals and the absence of distinct P waves [1][2].
Stroke Risk Scores: CHA2DS2-VASc vs. CHA2DS2-VA
For over a decade, the CHA2DS2-VASc score has been the foundational tool for assessing thromboembolic risk. However, recent epidemiological data have prompted a divergence in international guidelines regarding the role of biological sex. The 2024 European Society of Cardiology (ESC) guidelines formally updated the acronym to CHA2DS2-VA, explicitly removing female sex as an independent risk factor [2]. This change reflects robust evidence that female sex alone, in the absence of other comorbidities, does not significantly increase stroke risk. Conversely, the 2023 ACC/AHA guidelines retain the “VASc” nomenclature but clarify that female sex functions as a “risk modifier” rather than a primary driver of anticoagulation decisions [1]. In both paradigms, a score of ≥2 (excluding sex) is a Class I indication for OAC.
Bleeding Risk Assessment: HAS-BLED Score
Bleeding risk must be quantified using validated tools, with the HAS-BLED score being the most widely recommended. It evaluates Hypertension, Abnormal renal/liver function, Stroke history, Bleeding history, Labile INRs, Elderly age (>65), and Drugs/alcohol. A crucial clinical pitfall is the misapplication of this score. A high HAS-BLED score (≥3) indicates a need for closer clinical monitoring and aggressive modification of reversible risk factors (e.g., treating uncontrolled hypertension, discontinuing unnecessary NSAIDs or antiplatelets, and counseling on alcohol cessation). It is not a contraindication to OAC. Guidelines strongly discourage withholding anticoagulation based solely on a high bleeding risk score, as the net clinical benefit of stroke prevention almost universally outweighs the bleeding risk in high-CHA2DS2-VASc patients [1][3].
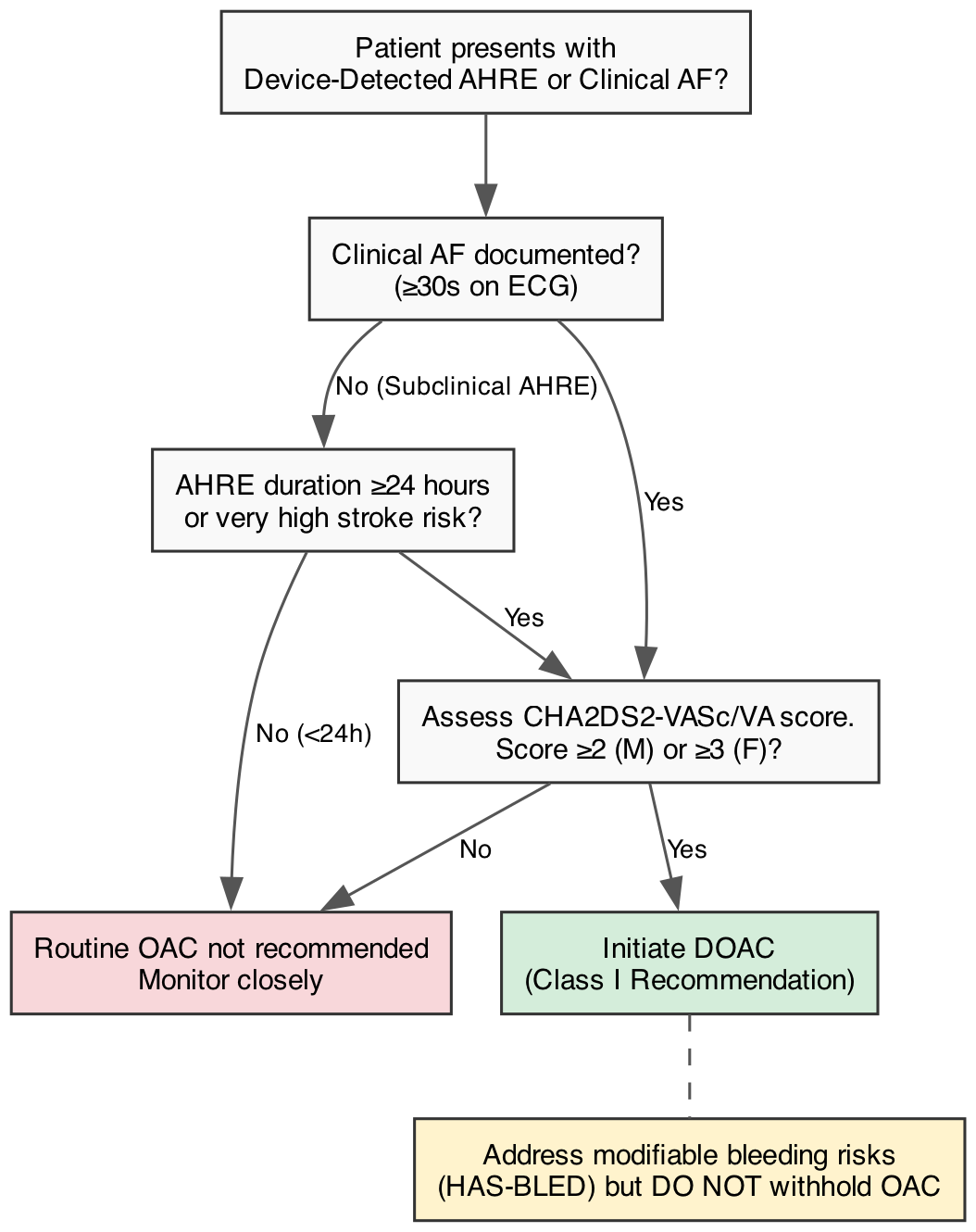
The Dilemma of Subclinical AF and AHREs
The proliferation of cardiac implantable electronic devices (CIEDs) and consumer wearables has introduced the diagnostic dilemma of Atrial High-Rate Episodes (AHREs) or subclinical AF. The management of these short-duration arrhythmias was recently clarified by two landmark trials: NOAH-AFNET 6 [24] and ARTESiA [23]. Both trials evaluated the use of Direct Oral Anticoagulants (DOACs) in patients with device-detected AHREs who lacked clinically documented AF. The consensus from both trials is that while DOACs reduce the incidence of ischemic stroke in this population, they simultaneously cause a significant increase in major bleeding events. Consequently, routine OAC is not recommended for short AHREs (<24 hours) unless the patient possesses an exceptionally high baseline stroke risk or transitions to clinically overt AF [2].
Foundational Pharmacotherapy: DOACs vs. Vitamin K Antagonists
For the prevention of stroke and systemic embolism in patients with non-valvular AF, DOACs (apixaban, dabigatran, edoxaban, and rivaroxaban) have supplanted Vitamin K Antagonists (VKAs, e.g., warfarin) as the Class I recommended first-line therapy [1][2]. This paradigm shift is underpinned by four pivotal randomized controlled trials (RCTs) and subsequent meta-analyses.
- RE-LY (Dabigatran): Demonstrated that dabigatran 150 mg twice daily was superior to warfarin for stroke prevention with similar major bleeding, while the 110 mg dose was non-inferior for stroke with significantly lower bleeding [5].
- ROCKET AF (Rivaroxaban): Showed that once-daily rivaroxaban was non-inferior to warfarin for the prevention of stroke and systemic embolism, though with a higher incidence of gastrointestinal bleeding [6].
- ARISTOTLE (Apixaban): Established that apixaban was superior to warfarin in preventing stroke, produced significantly less major bleeding, and resulted in lower all-cause mortality [7].
- ENGAGE AF-TIMI 48 (Edoxaban): Confirmed that edoxaban was non-inferior to warfarin for stroke prevention while significantly reducing major bleeding and cardiovascular death [8].
A comprehensive meta-analysis of these trials by Ruff et al. confirmed that DOACs, as a class, offer a favorable risk-benefit profile compared to warfarin. Most notably, DOACs reduce the risk of intracranial hemorrhage (ICH)—the most feared complication of anticoagulation—by approximately 50%, a finding that fundamentally altered the risk calculus of AF management [9].
Comparative Safety and Efficacy Among DOACs
While DOACs are universally preferred over VKAs for non-valvular AF, the absence of large, randomized head-to-head trials comparing individual DOACs complicates drug selection. Consequently, cardiologists must rely on robust, large-scale, real-world observational data to guide clinical choices.
The ARISTOPHANES study, a massive observational pooled analysis, provided critical insights into comparative effectiveness. It demonstrated that apixaban and dabigatran were associated with lower rates of major bleeding compared to warfarin, whereas rivaroxaban was associated with a higher rate of major bleeding [10].
Further granularity was provided by a landmark Medicare cohort study by Ray et al., which directly compared apixaban and rivaroxaban. The study revealed that apixaban was associated with a significantly lower risk of both major ischemic events and major hemorrhagic events (including gastrointestinal bleeding) compared to rivaroxaban [11]. These findings were corroborated by Fralick et al., reinforcing the preference for apixaban in routine practice, particularly for patients at elevated risk for gastrointestinal hemorrhage [12]. Edoxaban also demonstrates a favorable bleeding profile and serves as a viable alternative, though its use requires careful dose adjustment based on renal clearance and body weight [8].
Procedural and Peri-Procedural Management
Left Atrial Appendage Occlusion (LAAO)
For patients with AF who face absolute contraindications to long-term OAC (e.g., recurrent severe gastrointestinal bleeding or prior spontaneous ICH), percutaneous LAAO devices (e.g., Watchman, Amulet) offer a Class IIa indicated alternative. Landmark trials such as PROTECT AF, PREVAIL, and PRAGUE-17 established that percutaneous LAAO is non-inferior to OAC for stroke prevention, although the procedures carry inherent periprocedural risks, including pericardial effusion and device-related thrombosis [19][20][21].
In the realm of surgical management, the LAAOS III trial represented a watershed moment. It demonstrated a 33% reduction in ischemic stroke when the LAA was surgically closed during concomitant cardiac surgery in patients with AF, without increasing perioperative complications. This compelling data has elevated surgical LAAO to a Class I recommendation in both US and European guidelines for AF patients undergoing cardiac surgery for other indications [2][18].
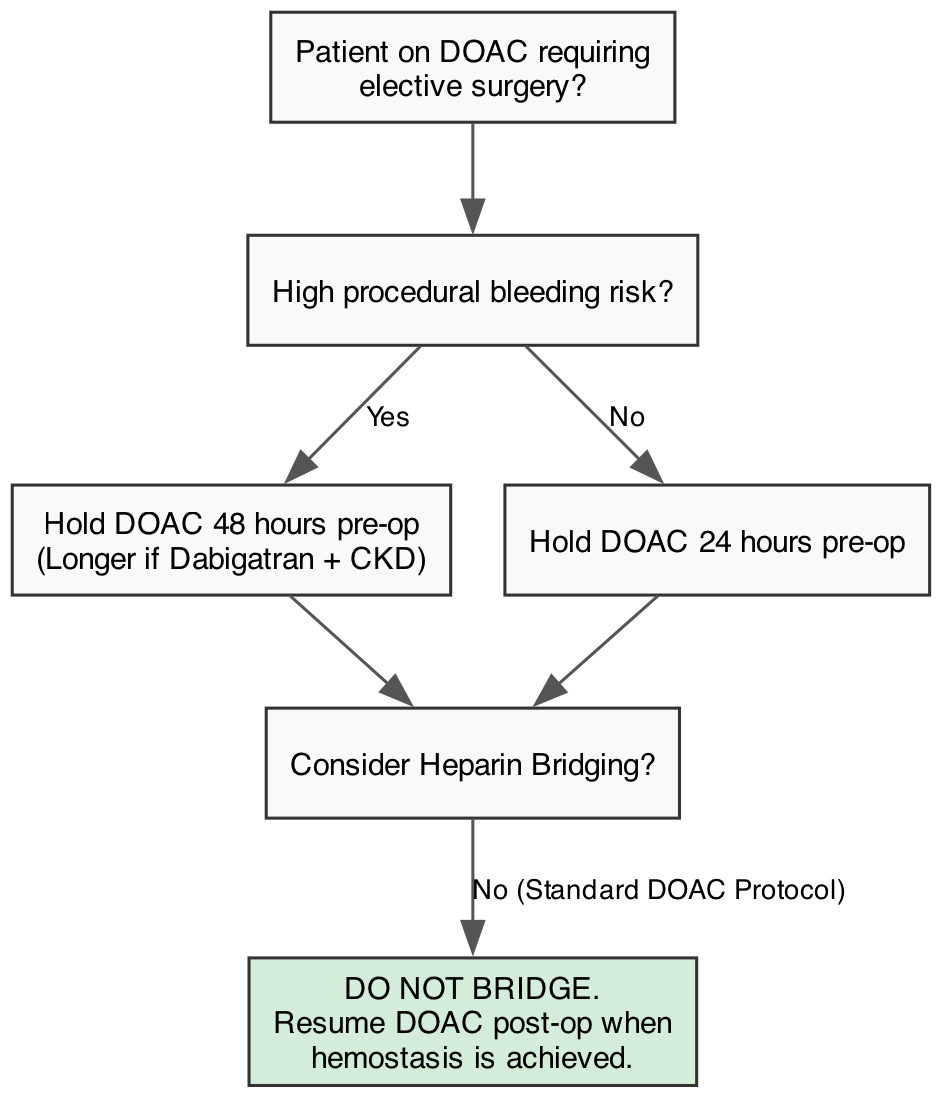
Peri-Procedural DOAC Management and the Fallacy of Bridging
The predictable pharmacokinetics and short half-lives of DOACs have simplified peri-procedural management. For elective surgeries, DOACs are typically held for 24 to 48 hours depending on the patient’s renal function and the procedural bleeding risk. Dabigatran, being 80% renally cleared, requires longer interruption times in patients with chronic kidney disease (CKD) [4].
A critical paradigm shift in contemporary cardiology is the near-total abandonment of heparin “bridging” for DOAC interruptions. Evidence unequivocally shows that bridging increases the risk of major bleeding without conferring any additional protection against stroke. Even for patients on VKAs, bridging is now heavily restricted and reserved almost exclusively for those with high-risk mechanical heart valves [4][1].
Special Populations and Clinical Controversies
Valvular AF: Rheumatic Heart Disease and Mechanical Valves
The term “non-valvular AF” is historically fraught, but contemporary guidelines are explicit: DOACs are strictly contraindicated in patients with moderate-to-severe rheumatic mitral stenosis and those with mechanical heart valves. The INVICTUS trial definitively demonstrated that VKAs are superior to rivaroxaban in preventing stroke and mortality in patients with rheumatic heart disease-associated AF [15][17]. Similarly, the PROACT Xa trial, which investigated apixaban in patients with mechanical On-X aortic valves, was halted early because the DOAC arm experienced significantly more thromboembolic events than the warfarin arm [16].
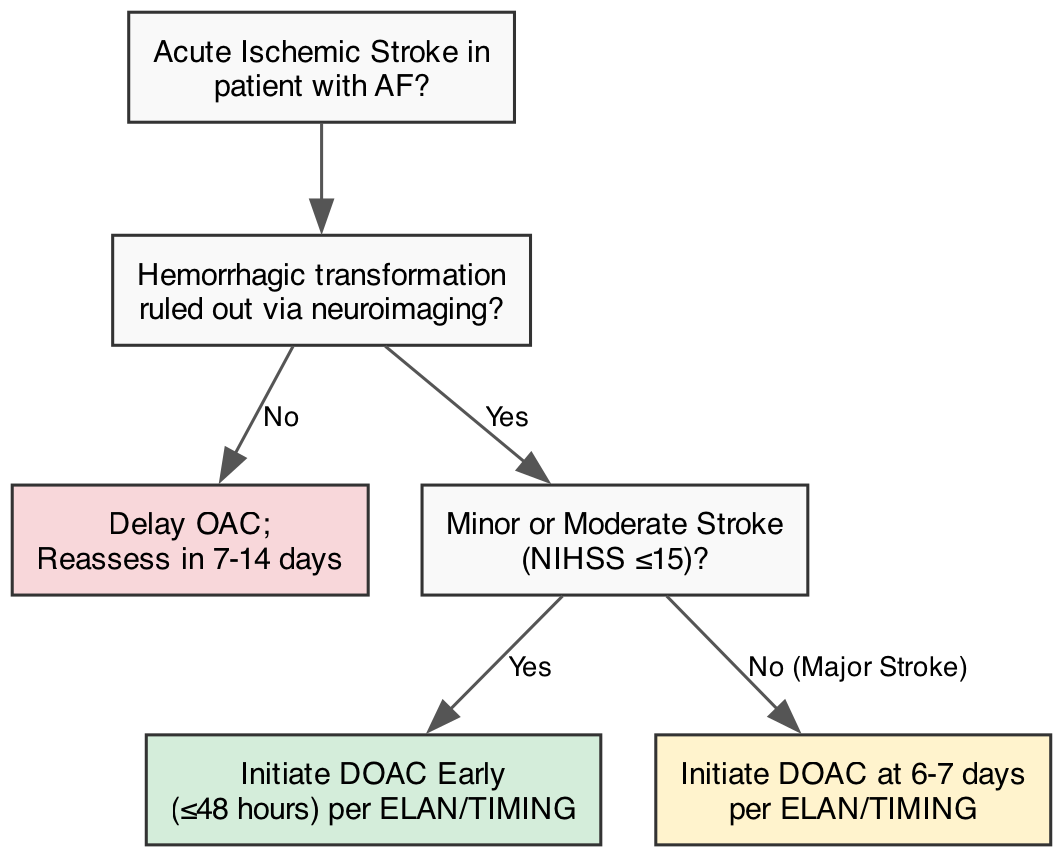
Post-Stroke Timing: The Shift to Early Initiation
Historically, restarting OAC after an acute ischemic stroke was delayed due to the fear of hemorrhagic transformation, governed by the empirical “1-3-6-12 day” rule based on stroke severity. Recent landmark RCTs have dismantled this dogma. The ELAN [13] and TIMING [14] trials demonstrated that early DOAC initiation (within 48 hours for minor/moderate stroke, or 6-7 days for major stroke) is safe, does not increase the rate of symptomatic ICH, and numerically reduces recurrent ischemic events compared to delayed initiation.
Chronic Kidney Disease (CKD) and End-Stage Renal Disease (ESRD)
All DOACs undergo renal clearance to varying degrees (dabigatran 80%, rivaroxaban 33%, apixaban 27%). In mild-to-moderate CKD, DOACs remain preferred over VKAs due to a superior safety profile. However, in ESRD patients on hemodialysis, management is highly controversial. In the United States, apixaban is frequently used off-label based on pharmacokinetic modeling and observational data suggesting safety [25]. RCT data remain mixed: the RENAL-AF trial (apixaban vs. warfarin) was underpowered and stopped early [26], while the AXADIA-AFNET 8 trial showed apixaban was non-inferior to VKAs for safety [27]. The VALKYRIE trial indicated that rivaroxaban reduced fatal/non-fatal cardiovascular events and major bleeding compared to VKAs in hemodialysis patients [28]. Despite these trials, a definitive Class I consensus for DOACs in ESRD remains elusive.
The Frail Elderly: The FRAIL-AF Caveat
While DOACs are generally considered safer than VKAs in the elderly, the recent FRAIL-AF trial introduced a critical caveat. The trial investigated switching frail older adults (≥75 years) who were already stable on a VKA to a DOAC. Surprisingly, the switch resulted in a significant increase in bleeding complications without any corresponding reduction in thromboembolic events [22]. Consequently, the 2024 ESC guidelines explicitly suggest avoiding the switch from a well-tolerated VKA to a DOAC in highly frail older adults, emphasizing that “if it isn’t broken, don’t fix it” [2].
Conclusions
The landscape of AF anticoagulation has been radically transformed over the past decade, moving from the monolithic use of warfarin to the nuanced, patient-specific application of DOACs and LAA occlusion devices. The refinement of risk scores (CHA2DS2-VA), the integration of wearable technology in diagnosing AHREs, and the paradigm shifts in post-stroke timing and peri-procedural management underscore a broader move toward precision cardiology.
Looking forward, the frontier of anticoagulation lies in the development of Factor XIa inhibitors (e.g., milvexian, asundexian), which promise to further uncouple hemostasis from thrombosis, potentially offering stroke prevention with near-zero bleeding risk. Until such therapies are validated in phase III trials, practicing cardiologists must continue to synthesize trial data, patient comorbidities, and anatomical considerations to optimize outcomes. Mastery of these strategies—recognizing when to initiate early post-stroke, when to avoid DOACs in valvular disease, and when to refer for LAAO—remains the hallmark of exceptional cardiovascular care.
- Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS Guideline for the Diagnosis and Management of Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149(1):e1-e156. DOI: 10.1161/CIR.0000000000001193
- Van Gelder IC, Rienstra M, Bunting KV, et al. 2024 ESC Guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2024;45(36):3314-3414. DOI: 10.1093/eurheartj/ehae176
- Hindricks G, Potpara T, Dagres N, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation. Eur Heart J. 2021;42(5):373-498. DOI: 10.1093/eurheartj/ehaa612
- Steffel J, Collins R, Antz M, et al. 2021 European Heart Rhythm Association Practical Guide on the Use of Non-Vitamin K Antagonist Oral Anticoagulants in Patients with Atrial Fibrillation. Europace. 2021;23(10):1612-1676. DOI: 10.1093/europace/euab065
- Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation (RE-LY). N Engl J Med. 2009;361(12):1139-1151. DOI: 10.1056/NEJMoa0905561
- Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation (ROCKET AF). N Engl J Med. 2011;365(10):883-891. DOI: 10.1056/NEJMoa1009638
- Granger CB, Alexander JH, McMurray JJ, et al. Apixaban versus warfarin in patients with atrial fibrillation (ARISTOTLE). N Engl J Med. 2011;365(11):981-992. DOI: 10.1056/NEJMoa1107039
- Giugliano RP, Ruff CT, Braunwald E, et al. Edoxaban versus warfarin in patients with atrial fibrillation (ENGAGE AF-TIMI 48). N Engl J Med. 2013;369(22):2093-2104. DOI: 10.1056/NEJMoa1310907
- Ruff CT, Giugliano RP, Braunwald E, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet. 2014;383(9921):955-962. DOI: 10.1016/S0140-6736(13)62343-0
- Lip GYH, Keshishian A, Kamble S, et al. Effectiveness and Safety of Oral Anticoagulants Among Nonvalvular Atrial Fibrillation Patients: The ARISTOPHANES Study. Stroke. 2018;49(12):2933-2944. DOI: 10.1161/STROKEAHA.118.022422
- Ray WA, Chung CP, Stein CM, et al. Association of Rivaroxaban vs Apixaban With Major Ischemic or Hemorrhagic Events in Patients With Atrial Fibrillation. JAMA. 2021;326(23):2395-2404. DOI: 10.1001/jama.2021.21222
- Fralick M, Colacci M, Schneeweiss S, et al. Effectiveness and Safety of Apixaban Compared With Rivaroxaban for Patients With Atrial Fibrillation in Routine Practice: A Cohort Study. Ann Intern Med. 2020;172(7):463-473. DOI: 10.7326/M19-2522
- Fischer U, Koga M, Strbian D, et al. Early versus Later Anticoagulation for Stroke with Atrial Fibrillation (ELAN). N Engl J Med. 2023;388(26):2411-2421. DOI: 10.1056/NEJMoa2303048
- Oldgren J, Åsberg S, Hijazi Z, et al. Early Versus Delayed Non-Vitamin K Antagonist Oral Anticoagulant Therapy After Acute Ischemic Stroke in Atrial Fibrillation (TIMING). Circulation. 2022;146(14):1056-1066. DOI: 10.1161/CIRCULATIONAHA.122.060666
- Connolly SJ, Karthikeyan G, Ntsekhe M, et al. Rivaroxaban in Rheumatic Heart Disease-Associated Atrial Fibrillation (INVICTUS). N Engl J Med. 2022;387(11):978-988. DOI: 10.1056/NEJMoa2209051
- Svensson LG, Wen J, et al. Apixaban or Warfarin in Patients with an On-X Mechanical Aortic Valve (PROACT Xa). NEJM Evid. 2023;2(7):EVIDoa2300067. DOI: 10.1056/EVIDoa2300067
- Weitz JI, Eikelboom JW, Connolly SJ. Warfarin faring better: vitamin K antagonists beat rivaroxaban and apixaban in the INVICTUS and PROACT Xa trials. J Thromb Haemost. 2023;21(11):3067-3071. DOI: 10.1016/j.jtha.2023.06.036
- Whitlock RP, Belley-Cote EP, Paparella D, et al. Left Atrial Appendage Occlusion during Cardiac Surgery to Prevent Stroke (LAAOS III). N Engl J Med. 2021;384(22):2081-2091. DOI: 10.1056/NEJMoa2101897
- Holmes DR, Reddy VY, Turi ZG, et al. Percutaneous closure of the left atrial appendage versus warfarin therapy for prevention of stroke in patients with atrial fibrillation (PROTECT AF). Lancet. 2009;374(9689):534-542. DOI: 10.1016/S0140-6736(09)61343-X
- Holmes DR Jr, Kar S, Price MJ, et al. Prospective randomized evaluation of the Watchman Left Atrial Appendage Closure device in patients with atrial fibrillation versus long-term warfarin therapy: the PREVAIL trial. J Am Coll Cardiol. 2014;64(1):1-12. DOI: 10.1016/j.jacc.2014.04.029
- Osmancik P, Herman D, Neuzil P, et al. Left Atrial Appendage Closure Versus Direct Oral Anticoagulants in High-Risk Patients With Atrial Fibrillation (PRAGUE-17). J Am Coll Cardiol. 2020;75(25):3122-3135. DOI: 10.1016/j.jacc.2020.04.067
- Joosten LPT, van Doorn S, van de Ven PM, et al. Safety of Switching from a Vitamin K Antagonist to a Non-Vitamin K Antagonist Oral Anticoagulant in Frail Older Patients with Atrial Fibrillation: Results of the FRAIL-AF Randomized Controlled Trial. Circulation. 2024;149(4):279-289. DOI: 10.1161/CIRCULATIONAHA.123.066485
- Healey JS, Lopes RD, Granger CB, et al. Apixaban for Stroke Prevention in Subclinical Atrial Fibrillation (ARTESiA). N Engl J Med. 2024;390(2):107-117. DOI: 10.1056/NEJMoa2310234
- Kirchhof P, Toennis T, Goette A, et al. Anticoagulation with Edoxaban in Patients with Atrial High-Rate Episodes (NOAH-AFNET 6). N Engl J Med. 2023;389(13):1167-1179. DOI: 10.1056/NEJMoa2303062
- Siontis KC, Zhang X, Eckard A, et al. Outcomes Associated With Apixaban Use in Patients With End-Stage Kidney Disease and Atrial Fibrillation in the United States. Circulation. 2018;138(15):1519-1529. DOI: 10.1161/CIRCULATIONAHA.118.035418
- Pokorney SD, Chertow GM, Al-Khalidi HR, et al. Apixaban for Patients With Atrial Fibrillation on Hemodialysis: A Multicenter Randomized Controlled Trial (RENAL-AF). Circulation. 2022;146(23):1735-1745. DOI: 10.1161/CIRCULATIONAHA.121.054990
- Reinecke H, Engelbertz C, Bauersachs R, et al. A Randomized Controlled Trial Comparing Apixaban With the Vitamin K Antagonist Phenprocoumon in Patients on Chronic Hemodialysis: The AXADIA-AFNET 8 Study. Circulation. 2023;147(4):296-309. DOI: 10.1161/CIRCULATIONAHA.122.062779
- De Vriese AS, Caluwé R, Bilo L, et al. Multicenter Randomized Controlled Trial of Vitamin K Antagonist Replacement by Rivaroxaban with or without Vitamin K2 in Hemodialysis Patients with Atrial Fibrillation: the Valkyrie Study. J Am Soc Nephrol. 2020;31(1):186-196. DOI: 10.1681/ASN.2019060579

