Definition and terminology
Heart failure (HF) is a complex clinical syndrome characterized by symptoms and signs resulting from structural or functional impairment of ventricular filling or ejection of blood. The 2021 Universal Definition of Heart Failure, endorsed by major global cardiovascular societies, defines HF as a clinical syndrome corroborated by elevated natriuretic peptide levels and/or objective evidence of pulmonary or systemic congestion [3]. Within this spectrum, Heart Failure with Reduced Ejection Fraction (HFrEF) is specifically classified by the 2022 AHA/ACC/HFSA and 2021/2023 ESC guidelines as HF with a left ventricular ejection fraction (LVEF) of ≤40% [14, 26, 27].
Recent guideline updates have introduced critical nuances to HF terminology to reflect the dynamic nature of myocardial remodeling. The designation “Heart Failure with improved Ejection Fraction” (HFimpEF) is now applied to patients with a baseline LVEF ≤40% who have experienced a ≥10-point increase in LVEF, with a subsequent measurement of >40% [14]. This distinction is paramount for practicing cardiologists: HFimpEF represents a state of myocardial remission rather than true recovery. Data from the TRED-HF trial demonstrated that withdrawal of guideline-directed medical therapy (GDMT) in these patients leads to a high rate of relapse, underscoring the mandate that HFrEF pharmacotherapy must be continued indefinitely even after LVEF normalization.
Epidemiology and risk factors
The global burden of heart failure is staggering, affecting an estimated 64 million individuals worldwide. In developed nations, the prevalence of HF is approximately 1–2% in the general adult population, but this figure rises steeply with age, exceeding 10% in individuals over 70 years [48]. The lifetime risk of developing HF for an individual at age 40 is estimated at 24%, a figure that remains consistent across both men and women [10].
Registry data, such as those from the Framingham Heart Study and the Atherosclerosis Risk in Communities (ARIC) study, highlight a shifting demographic profile in incident HFrEF. Historically dominated by acute myocardial infarction (MI) survivors, the contemporary risk factor profile is increasingly driven by the global epidemics of obesity, diabetes mellitus, and metabolic syndrome [58]. Hypertension and coronary artery disease (CAD) remain the most potent population-attributable risk factors. Furthermore, the “obesity paradox”—whereby overweight and mildly obese HF patients exhibit better survival than normal-weight counterparts—remains a subject of intense epidemiological debate, likely reflecting the catabolic state of advanced HF (cardiac cachexia) rather than a protective effect of adiposity.
Etiology and underlying causes
The etiology of HFrEF is broadly dichotomized into ischemic and non-ischemic causes, a distinction that fundamentally alters diagnostic work-up and device therapy eligibility. Ischemic cardiomyopathy, defined by significant CAD (prior MI, diffuse microvascular dysfunction, or flow-limiting epicardial stenoses), is the predominant cause, accounting for approximately 60% of HFrEF cases in Western populations [14].
Non-ischemic etiologies encompass a highly heterogeneous group of disorders. Idiopathic dilated cardiomyopathy (DCM) is frequently diagnosed when ischemic, valvular, and hypertensive causes are excluded. However, advances in molecular diagnostics reveal that up to 30-40% of “idiopathic” cases harbor a genetic basis. Truncating variants in the TTN gene (titin) are the most common genetic cause, followed by mutations in LMNA, MYH7, and TNNT2 [39]. Consequently, current guidelines recommend a 3-generation family history and consideration of genetic testing for all patients with non-ischemic HFrEF.
Toxic and infiltrative causes are increasingly recognized. The burgeoning field of cardio-oncology has highlighted the cardiotoxic effects of anthracyclines, HER2 inhibitors (trastuzumab), and immune checkpoint inhibitors, which can precipitate fulminant myocarditis and subsequent HFrEF [22]. Substance abuse (alcohol, methamphetamines) and infiltrative diseases (cardiac amyloidosis, sarcoidosis) must also be systematically evaluated, as they require specific, targeted interventions.
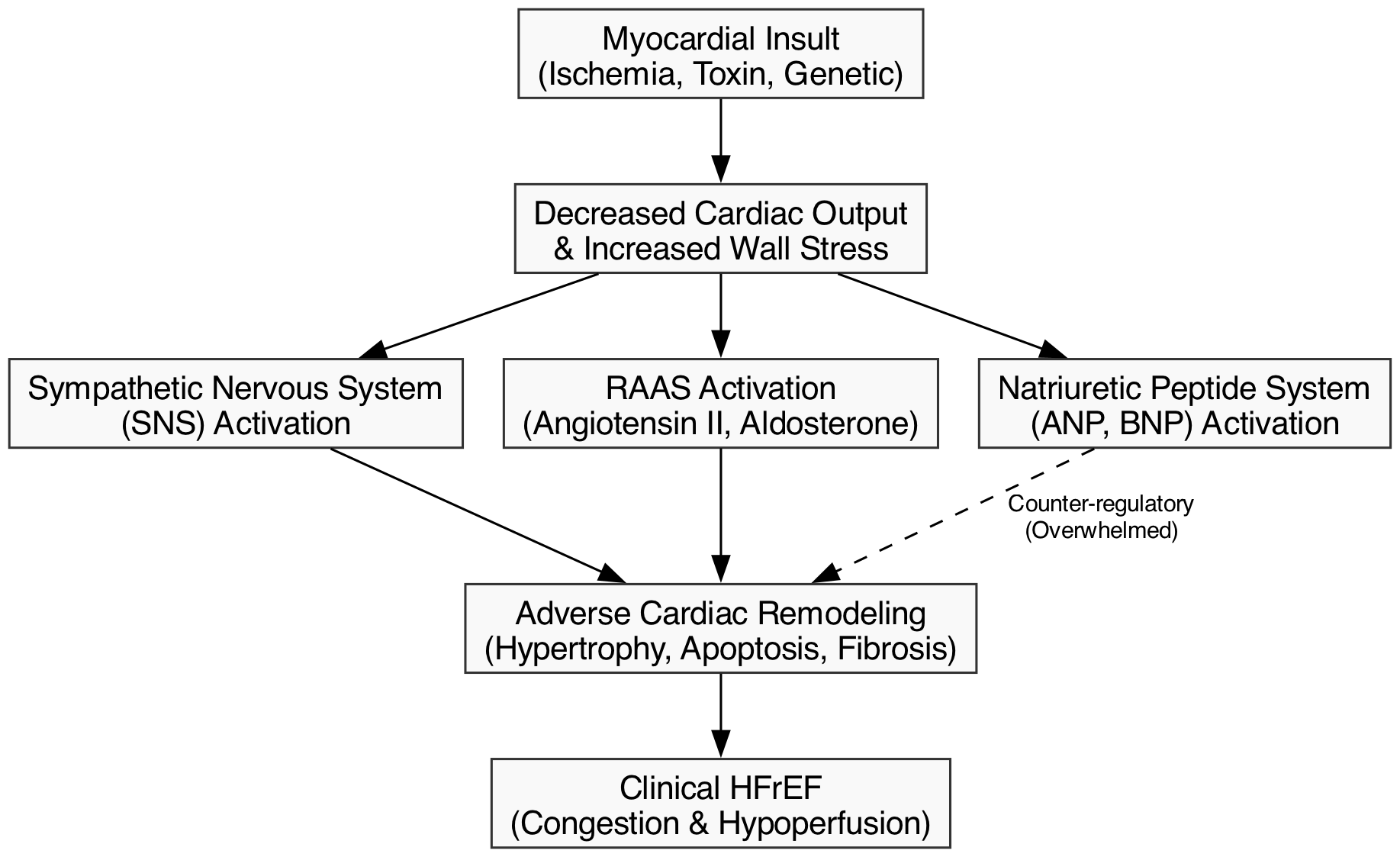
Pathophysiology and cardiac remodeling
The transition from an initial myocardial insult to chronic HFrEF is governed by the neurohormonal hypothesis. An initial decline in cardiac output triggers a compensatory, yet ultimately maladaptive, chronic activation of the sympathetic nervous system (SNS) and the renin-angiotensin-aldosterone system (RAAS) [40]. While these systems acutely maintain perfusion via vasoconstriction and sodium retention, their chronic activation exerts profound toxic effects on the myocardium.
At the cellular level, chronic neurohormonal stimulation induces myocyte hypertrophy, apoptosis, and fibroblast proliferation, leading to extracellular matrix deposition and interstitial fibrosis. This process, termed adverse cardiac remodeling, manifests macroscopically as the left ventricle (LV) transitioning from a prolate ellipse to a spherical shape. This spherical dilation increases wall stress (governed by Laplace’s law), exacerbates subendocardial ischemia, and causes papillary muscle displacement, leading to secondary (functional) mitral regurgitation [24].
Counter-regulatory systems, primarily the natriuretic peptide system (ANP, BNP), attempt to mitigate this by inducing vasodilation, natriuresis, and anti-fibrotic effects. However, in chronic HFrEF, this endogenous defense is overwhelmed by the RAAS/SNS axis. Modern pharmacotherapy, particularly neprilysin inhibition, is specifically designed to augment these beneficial endogenous peptides while simultaneously blocking the maladaptive RAAS pathways [4].
Clinical presentation
The clinical presentation of HFrEF is a manifestation of two primary hemodynamic derangements: backward failure (venous congestion) and forward failure (systemic hypoperfusion). Patients are often categorized using the Stevenson hemodynamic profiles: Warm and Dry (compensated), Warm and Wet (congested, most common), Cold and Dry (hypoperfused), and Cold and Wet (cardiogenic shock) [47].
Symptoms of backward failure include exertional dyspnea, orthopnea, paroxysmal nocturnal dyspnea (PND), lower extremity edema, and abdominal fullness due to congestive hepatopathy and gut edema. Forward failure presents as profound fatigue, weakness, altered mental status, and cool extremities [26].
The physical examination remains a cornerstone of clinical assessment. Elevated jugular venous pressure (JVP) and a positive hepatojugular reflux (HJR) are highly reliable markers of volume overload. The presence of a third heart sound (S3 gallop) is highly specific for elevated left ventricular end-diastolic pressure and impending decompensation. While pulmonary crackles are a classic sign of acute pulmonary edema, they are frequently absent in chronic HFrEF due to compensatory increases in lymphatic drainage [59].
Diagnostic criteria and work-up
The diagnostic work-up for suspected HFrEF aims to confirm the diagnosis, establish the etiology, and identify precipitating factors. The initial evaluation must include a comprehensive history and physical examination, a 12-lead electrocardiogram (ECG), and a chest radiograph [14]. The ECG is rarely normal in HFrEF; the presence of Q waves suggests prior MI, while a left bundle branch block (LBBB) indicates intraventricular conduction delay, a critical factor for device therapy planning. The chest X-ray may reveal cardiomegaly, cephalization of pulmonary vessels, and pleural effusions, though its sensitivity is limited.
Given that CAD is the leading cause of HFrEF, an ischemic evaluation is mandatory for all patients presenting with new-onset HFrEF without a clear non-ischemic etiology. Coronary angiography remains the gold standard, but coronary computed tomography angiography (CCTA) is an increasingly utilized Class IIa alternative for patients with a low-to-intermediate pre-test probability of CAD [14].

Echocardiography and imaging
Transthoracic echocardiography (TTE) is the undisputed Class 1 gold standard for the initial structural and functional assessment of the failing heart. TTE provides critical data on LVEF (preferably calculated via the Simpson’s biplane method), chamber dimensions, wall thickness, and diastolic function. Furthermore, it is essential for evaluating valvular hemodynamics, particularly the severity of secondary mitral and tricuspid regurgitation, and for estimating non-invasive right ventricular systolic pressures [20].
When TTE windows are inadequate or when precise tissue characterization is required, Cardiac Magnetic Resonance (CMR) imaging is strongly recommended. CMR is the gold standard for volumetric quantification. More importantly, late gadolinium enhancement (LGE) imaging allows for the differentiation of ischemic versus non-ischemic etiologies. Ischemic cardiomyopathy typically presents with subendocardial or transmural LGE in a coronary distribution, whereas non-ischemic DCM often shows mid-wall or epicardial striae. CMR is also indispensable for diagnosing infiltrative cardiomyopathies such as cardiac amyloidosis and sarcoidosis [15].
Biomarkers and laboratory assessment
Laboratory assessment is vital for identifying comorbidities and guiding therapy. Routine tests include a complete blood count, comprehensive metabolic panel, lipid profile, HbA1c, and thyroid-stimulating hormone (TSH). Iron studies (ferritin and transferrin saturation) are now mandatory due to the prognostic and therapeutic implications of iron deficiency in HFrEF [26].
B-type natriuretic peptide (BNP) and N-terminal pro-B-type natriuretic peptide (NT-proBNP) are the premier biomarkers for diagnosis and prognostication. For non-acute presentations, the rule-out thresholds are BNP <35 pg/mL or NT-proBNP <125 pg/mL. In the acute setting, thresholds are higher (BNP <100 pg/mL or NT-proBNP <300 pg/mL) [16, 37]. It is critical to note that neprilysin inhibitors (ARNI) degrade BNP but not NT-proBNP; thus, NT-proBNP is the preferred biomarker for monitoring patients on sacubitril/valsartan.
High-sensitivity cardiac troponin (hs-cTn) is useful for detecting acute myocardial injury as a precipitant of decompensation and provides independent prognostic value [37]. The recent STRONG-HF trial demonstrated that a biomarker-guided approach—using NT-proBNP levels to safely guide the rapid up-titration of GDMT—significantly improves clinical outcomes, cementing the role of serial biomarker evaluation in routine practice [30].
Staging and classification (ACC/AHA, NYHA)
The ACC/AHA stages of heart failure emphasize the progressive nature of the disease and the importance of early intervention [14]:
- Stage A: At risk for HF (e.g., hypertension, diabetes, obesity) but without structural heart disease or symptoms.
- Stage B: Pre-HF; structural heart disease (e.g., LV hypertrophy, prior MI) or elevated biomarkers, but no symptoms.
- Stage C: Symptomatic HF; prior or current symptoms of HF associated with structural heart disease.
- Stage D: Advanced HF; severe symptoms at rest, recurrent hospitalizations despite maximal GDMT, requiring specialized interventions.
Importantly, the ACC/AHA staging is unidirectional; a patient cannot regress from Stage C to Stage B, even if symptoms resolve. In contrast, the New York Heart Association (NYHA) functional classification gauges current symptom severity and can fluctuate [6]:
- Class I: No limitation of physical activity.
- Class II: Slight limitation; ordinary activity causes symptoms.
- Class III: Marked limitation; less than ordinary activity causes symptoms.
- Class IV: Inability to carry out any physical activity without discomfort; symptoms present at rest.
Guideline-directed medical therapy (GDMT)
The cornerstone of HFrEF management has undergone a radical paradigm shift. The 2022 AHA/ACC and 2023 ESC guidelines mandate the use of quadruple therapy—often termed the “Four Pillars”—for all patients with HFrEF. These include: (1) an ARNI (or ACEi/ARB), (2) an evidence-based beta-blocker, (3) a mineralocorticoid receptor antagonist (MRA), and (4) an SGLT2 inhibitor. All four classes carry a Class 1 recommendation [14, 27].
Historically, GDMT was initiated in a stepwise, sequential manner, often taking months to achieve comprehensive blockade. Contemporary guidelines and consensus pathways now strongly advocate for simultaneous or rapid sequential initiation of all four pillars at low doses within weeks of diagnosis. This approach is driven by evidence that the morbidity and mortality benefits of these agents are additive, independent, and occur rapidly (within 2-4 weeks of initiation) [23, 49].

Renin–angiotensin system inhibition (ACEi/ARB/ARNI)
Inhibition of the RAAS is foundational. The angiotensin receptor-neprilysin inhibitor (ARNI), sacubitril/valsartan, is the preferred agent (Class 1a) over ACE inhibitors and ARBs. The landmark PARADIGM-HF trial demonstrated that ARNI therapy yielded a 20% relative risk reduction in cardiovascular death or HF hospitalization compared to enalapril, alongside significant improvements in quality of life [28]. The PIONEER-HF trial further established the safety and superior efficacy of initiating ARNI in-hospital during acute decompensation [57].
If an ARNI is unavailable, unaffordable, or poorly tolerated, ACE inhibitors (e.g., enalapril, lisinopril) or ARBs (e.g., candesartan, valsartan) remain Class 1 recommendations, backed by legacy trials such as SOLVD and CHARM-Alternative [11, 50]. Sub-group analyses from the PARAGON-HF trial (which studied HFpEF) suggest that the benefits of ARNI extend into the mildly reduced EF range (HFmrEF), particularly in women, prompting broader utilization across the LVEF spectrum.
Beta-blockers
Beta-blockers mitigate the cardiotoxic effects of chronic sympathetic activation, reduce heart rate, decrease myocardial oxygen demand, and are highly effective at reversing LV remodeling and preventing sudden cardiac death. However, class effects do not apply; only three specific beta-blockers have proven mortality benefits in HFrEF: bisoprolol (CIBIS-II), sustained-release metoprolol succinate (MERIT-HF), and carvedilol (COPERNICUS) [7, 35, 42].
Beta-blockers should be initiated in clinically stable, euvolemic patients. In the setting of acute decompensated HF, established beta-blocker therapy should generally be continued at a reduced dose unless the patient is in cardiogenic shock or exhibits severe hypoperfusion, in which case temporary cessation is required.
Mineralocorticoid receptor antagonists
Aldosterone drives myocardial fibrosis, sodium retention, and endothelial dysfunction. The MRAs spironolactone and eplerenone block these deleterious effects. The RALES trial (spironolactone in severe HF) and the EMPHASIS-HF trial (eplerenone in mild HF) demonstrated profound reductions in all-cause mortality and HF hospitalizations [44, 60].
The primary barrier to MRA utilization is the risk of hyperkalemia and renal dysfunction, particularly when combined with an ARNI/ACEi/ARB. Careful surveillance of serum potassium and creatinine is mandatory. The recent DIAMOND trial demonstrated that the use of the novel potassium binder patiromer enables the optimization of RAAS inhibitors and MRAs in patients who would otherwise be intolerant due to hyperkalemia, offering a new strategy for maximizing GDMT.
SGLT2 inhibitors
Sodium-glucose cotransporter-2 (SGLT2) inhibitors represent the most significant breakthrough in HF pharmacotherapy in the last decade. Originally developed as anti-hyperglycemic agents, dapagliflozin and empagliflozin are now foundational HFrEF therapies regardless of the patient’s diabetes status.
The DAPA-HF and EMPEROR-Reduced trials showed approximately 25% relative risk reductions in cardiovascular death or worsening HF [29, 41]. The mechanisms of benefit are pleiotropic and independent of glycemic control, involving improved myocardial energetics (shift toward ketone utilization), reduction in interstitial edema via proximal tubule natriuresis, and inhibition of the Na+/H+ exchanger (NHE1). Meta-analyses of pooled data confirm robust reductions in all-cause mortality and highlight profound renal protective effects, slowing the progression of chronic kidney disease (CKD) in this vulnerable population [55].
Additional pharmacologic therapies (ivabradine, hydralazine–nitrates, digoxin)
Beyond the four pillars, several adjunctive therapies are indicated for specific clinical scenarios:
- Ivabradine: This agent selectively inhibits the $I_f$ channel in the sinoatrial node, reducing heart rate without negative inotropic effects. Based on the SHIFT trial, it is indicated for symptomatic HFrEF patients with LVEF ≤35%, in sinus rhythm, with a resting HR ≥70 bpm despite maximally tolerated beta-blockers [52].
- Hydralazine and Isosorbide Dinitrate: This combination provides a Class 1 recommendation for self-identified African American patients with NYHA class III–IV HFrEF who remain symptomatic despite optimal GDMT, based on the survival benefit demonstrated in the A-HeFT trial [53].
- Vericiguat: An oral soluble guanylate cyclase (sGC) stimulator that enhances the cyclic GMP pathway. The VICTORIA trial showed that vericiguat reduces the composite of CV death or HF hospitalization in high-risk patients with worsening HF (recent hospitalization or need for IV diuretics) [1].
- Digoxin: While it provides no mortality benefit, digoxin may be considered for symptom control and reduction of HF hospitalizations in patients who remain symptomatic on maximal GDMT, as shown in the DIG trial [9].
Diuretic therapy and volume management
Loop diuretics (furosemide, bumetanide, torsemide) are the mainstay for alleviating congestion. Despite theoretical pharmacokinetic advantages of torsemide, the recent pragmatic TRANSFORM-HF trial showed no significant difference in all-cause mortality or hospitalization between torsemide and furosemide [33].
Diuretic resistance is a frequent clinical challenge, often mediated by distal nephron hypertrophy. Sequential nephron blockade is the preferred strategy. The ADVOR trial demonstrated that the addition of intravenous acetazolamide (a proximal tubule carbonic anhydrase inhibitor) to loop diuretics significantly improved the incidence of successful decongestion compared to placebo [38]. Furthermore, the PUSH-AHF trial validated a natriuresis-guided approach, showing that using early spot urine sodium measurements to guide IV diuretic dosing improves natriuresis and clinical decongestion [54].

Optimization and titration of therapy
Initiation of GDMT is only the first step; optimization to target doses is where the maximum survival benefit is realized. Registry data, such as the CHAMP-HF registry, reveal pervasive clinical inertia, with less than 25% of eligible patients achieving target doses of GDMT [12].
Guidelines stress that target doses derived from randomized controlled trials must be pursued, rather than stopping titration once symptom relief is achieved. The STRONG-HF trial provided a definitive mandate for high-intensity care: rapid up-titration of GDMT to 100% of recommended doses within 2 weeks of hospital discharge, guided by NT-proBNP levels and close clinical follow-up, significantly reduced 180-day HF readmission or death compared to usual care [30].
Management of comorbidities
Comorbidities frequently complicate HFrEF management and drive hospitalizations.
- Iron Deficiency: Present in up to 50% of HFrEF patients, iron deficiency is defined as ferritin <100 µg/L or 100–299 µg/L with transferrin saturation (TSAT) <20%. Intravenous iron repletion (ferric carboxymaltose or ferric derisomaltose) improves symptoms, exercise capacity, and quality of life. The AFFIRM-AHF and IRONMAN trials demonstrated reductions in HF hospitalizations [18, 46]. The recent HEART-FID trial showed marginal but safe results for FCM, cementing IV iron as a standard of care for symptomatic patients [34].
- Atrial Fibrillation (AF): AF and HF frequently coexist and exacerbate one another. The CASTLE-AF trial demonstrated that catheter ablation is superior to medical therapy for rhythm control in select HFrEF patients, significantly improving survival and LVEF [25].
- Chronic Kidney Disease (CKD): Cardiorenal syndrome is ubiquitous. SGLT2 inhibitors are highly effective at slowing CKD progression in HFrEF patients, as evidenced by the DAPA-CKD and EMPA-KIDNEY trials, and should be continued even as GFR declines to 20 mL/min/1.73m² [13].
Device therapy (ICD, CRT)
Device therapy is indicated when structural and electrical remodeling place the patient at high risk for sudden cardiac death (SCD) or pump failure despite optimal GDMT.
Implantable Cardioverter-Defibrillator (ICD): ICDs carry a Class 1 recommendation for the primary prevention of SCD in patients with an LVEF ≤35% and NYHA II-III symptoms despite ≥3 months of GDMT, with an expected survival of >1 year. This is based on landmark trials like MADIT-II (ischemic) and SCD-HeFT (mixed etiology) [2, 36]. However, the DANISH trial introduced nuance, showing a less clear all-cause mortality benefit for prophylactic ICDs in purely non-ischemic etiologies, though SCD was reduced [19].
Cardiac Resynchronization Therapy (CRT): CRT is indicated for patients with LVEF ≤35%, sinus rhythm, and a Left Bundle Branch Block (LBBB) with a QRS duration ≥150 ms. By restoring electromechanical synchrony, CRT improves symptoms, induces reverse remodeling, and reduces mortality, as demonstrated in the COMPANION and CARE-HF trials [5, 8].

Revascularization and surgical options
The role of revascularization in ischemic cardiomyopathy depends heavily on the modality and the presence of viable myocardium. Coronary artery bypass grafting (CABG) is recommended for patients with multivessel CAD and HFrEF. The 10-year follow-up of the STICH trial demonstrated a significant all-cause mortality benefit for CABG over medical therapy alone, highlighting the long-term protective effect of surgical revascularization against future ischemic events [56].
Conversely, the role of percutaneous coronary intervention (PCI) in stable ischemic LV dysfunction was recently challenged by the REVIVED-BCIS2 trial. This landmark study showed that PCI did not reduce all-cause mortality or HF hospitalization compared to optimal medical therapy alone, suggesting that stable ischemic HFrEF should be managed medically unless the patient has refractory angina [43].
For patients with severe secondary (functional) mitral regurgitation who remain symptomatic despite optimal GDMT and CRT, Transcatheter Edge-to-Edge Repair (TEER, e.g., MitraClip) is a viable option. The COAPT trial demonstrated that TEER significantly reduces hospitalizations and mortality in appropriately selected patients with disproportionate mitral regurgitation [51].

Advanced heart failure therapies (LVAD, transplantation)
Patients progressing to Stage D HF—characterized by recurrent hospitalizations, intolerance to GDMT due to hypotension or renal failure, or the need for inotropic support—must be referred to an advanced HF center. The “I-NEED-HELP” mnemonic is a useful trigger for timely referral.
For eligible candidates, orthotopic heart transplantation remains the gold standard, offering a median survival exceeding 12 years [31]. However, due to donor organ shortages, Left Ventricular Assist Devices (LVADs) are increasingly utilized as both a bridge to transplant and as destination therapy. The MOMENTUM 3 trial established the superiority of the HeartMate 3—a fully magnetically levitated continuous-flow pump—which drastically reduced the incidence of pump thrombosis and stroke compared to older axial-flow devices, making LVAD therapy a durable long-term option [32].

Prognosis
Prognostication in HFrEF is essential for guiding patient expectations and timing advanced therapies. Validated multivariable risk models, such as the MAGGIC risk score and the Seattle Heart Failure Model (SHFM), are recommended to estimate 1-year and 3-year survival [21, 45].
Historically, the 5-year mortality for HFrEF hovered around 50%. However, the contemporary landscape has been drastically altered by comprehensive GDMT. Modeling studies indicate that the optimal implementation of all four pillars (ARNI, beta-blocker, MRA, SGLT2i) extends survival by an estimated 6 to 8 years in a 55-year-old patient compared to conventional therapy (ACEi and beta-blocker alone) [55].
Ongoing Trials and Future Directions
The therapeutic horizon for HFrEF continues to expand. Ongoing trials are investigating novel pathways, such as cardiac myosin activators (following the mixed results of omecamtiv mecarbil in GALACTIC-HF), GLP-1 receptor agonists for obesity-related HF phenotypes, and gene therapies targeting calcium handling (SERCA2a) and specific genetic cardiomyopathies (e.g., CRISPR for TTR amyloidosis and MYBPC3 mutations). Furthermore, the optimization of HFimpEF management remains a fertile ground for research, with trials aiming to identify which patients might safely de-escalate therapy without risking relapse. As the armamentarium grows, the focus of practicing cardiologists must remain on overcoming clinical inertia and ensuring equitable delivery of life-saving, guideline-directed therapies to all patients.
- Armstrong PW, Pieske B, Anstrom KJ, et al. Vericiguat in Patients with Heart Failure and Reduced Ejection Fraction. N Engl J Med. 2020;382:1883-1893. DOI: 10.1056/NEJMoa1915928
- Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med. 2005;352:225-237. DOI: 10.1056/NEJMoa043399
- Bozkurt B, Coats AJS, Tsutsui H, et al. Universal definition and classification of heart failure. J Card Fail. 2021;27(4):387-413. DOI: 10.1016/j.cardfail.2021.01.022
- Braunwald E. The war against heart failure: the Lancet lecture. Lancet. 2015;385(9970):812-824. DOI: 10.1016/S0140-6736(14)61889-4
- Bristow MR, Saxon LA, Boehmer J, et al. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N Engl J Med. 2004;350:2140-2150. DOI: 10.1056/NEJMoa032423
- Caraballo C, Desai NR, Mulder H, et al. Clinical Implications of the New York Heart Association Classification. J Am Heart Assoc. 2019;8(23):e014240. DOI: 10.1161/JAHA.119.014240
- CIBIS-II Investigators. The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): a randomised trial. Lancet. 1999;353:9-13. DOI: 10.1016/S0140-6736(98)11181-9
- Cleland JGF, Daubert JC, Erdmann E, et al. The effect of cardiac resynchronization on morbidity and mortality in heart failure. N Engl J Med. 2005;352:1539-1549. DOI: 10.1056/NEJMoa050496
- DIG Trial Investigators. The effect of digoxin on mortality and morbidity in patients with heart failure. N Engl J Med. 1997;336:525-533. DOI: 10.1056/NEJM199702203360801
- Emmons-Bell S, Johnson C, Roth G. Prevalence, incidence and survival of heart failure: a systematic review. Heart. 2022;108:1351-1360. DOI: 10.1136/heartjnl-2021-320136
- Granger CB, McMurray JJV, Yusuf S, et al. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function intolerant to angiotensin-converting-enzyme inhibitors: the CHARM-Alternative trial. Lancet. 2003;362:772-776. DOI: 10.1016/S0140-6736(03)14284-5
- Greene SJ, Butler J, Albert NM, et al. Medical Therapy for Heart Failure With Reduced Ejection Fraction: The CHAMP-HF Registry. J Am Coll Cardiol. 2021;72:351-366. DOI: 10.1016/j.jacc.2018.04.070
- Heerspink HJL, Stefánsson BV, Correa-Rotter R, et al. Dapagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2020;383:1436-1446. DOI: 10.1056/NEJMoa2024816
- Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure. J Am Coll Cardiol. 2022;79(17):e263-e421. DOI: 10.1016/j.jacc.2021.12.012
- Hundley WG, Bluemke DA, Finn JP, et al. ACCF/ACR/AHA/NASCI/SCMR 2010 expert consensus document on cardiovascular magnetic resonance. J Am Coll Cardiol. 2010;55:2614-2662. DOI: 10.1016/j.jacc.2009.11.011
- Januzzi JL, Ahmad T, Celano CM, et al. Core Biomarker Testing in Heart Failure. Circulation. 2018;138:e19-e26. DOI: 10.1161/CIR.0000000000000589
- Jones NR, Roalfe AK, Adoki I, et al. Survival of patients with chronic heart failure in the community: a systematic review and meta-analysis. Eur J Heart Fail. 2019;21:1306-1325. DOI: 10.1002/ejhf.1594
- Kalra PR, Cleland JGF, Petrie MC, et al. Intravenous ferric derisomaltose in patients with heart failure and iron deficiency in the UK (IRONMAN). Lancet. 2022;400(10369):2199-2209. DOI: 10.1016/S0140-6736(22)02083-9
- Kober L, Thune JJ, Nielsen JC, et al. Defibrillator Implantation in Patients with Nonischemic Systolic Heart Failure. N Engl J Med. 2016;375:1221-1230. DOI: 10.1056/NEJMoa1608029
- Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults. J Am Soc Echocardiogr. 2015;28:1-39. DOI: 10.1016/j.echo.2014.10.003
- Levy WC, Mozaffarian D, Linker DT, et al. The Seattle Heart Failure Model: prediction of survival in heart failure. Circulation. 2006;113:1424-1433. DOI: 10.1161/CIRCULATIONAHA.105.584102
- Lyon AR, López-Fernández T, Couch LS, et al. 2022 ESC Guidelines on cardio-oncology. Eur Heart J. 2022;43(41):4229-4361. DOI: 10.1093/eurheartj/ehac244
- Maddox TM, Januzzi JL, Allen LA, et al. 2021 Update to the 2017 ACC Expert Consensus Decision Pathway for Optimization of Heart Failure Treatment. J Am Coll Cardiol. 2021;77(6):772-810. DOI: 10.1016/j.jacc.2020.11.022
- Mann DL. Pathophysiology of heart failure. In: Braunwald’s Heart Disease. 9th ed. 2012.
- Marrouche NF, Brachmann J, Andresen D, et al. Catheter Ablation for Atrial Fibrillation with Heart Failure. N Engl J Med. 2018;378:417-427. DOI: 10.1056/NEJMoa1707855
- McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599-3726. DOI: 10.1093/eurheartj/ehab368
- McDonagh TA, Metra M, Adamo M, et al. 2023 ESC Guidelines for the management of heart failure focused update. Eur Heart J. 2023;44(32):3028-3114. DOI: 10.1093/eurheartj/ehad195
- McMurray JJV, Packer M, Desai AS, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371:993-1004. DOI: 10.1056/NEJMoa1409077
- McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N Engl J Med. 2019;381:1995-2008. DOI: 10.1056/NEJMoa1911303
- Mebazaa A, Davison B, Chioncel O, et al. Safety, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute heart failure (STRONG-HF): a multinational, open-label, randomised, trial. Lancet. 2022;400(10367):1938-1952. DOI: 10.1016/S0140-6736(22)02076-1
- Mehra MR, Canter CE, Hannan MM, et al. The 2016 International Society for Heart Lung Transplantation listing criteria for heart transplantation. J Heart Lung Transplant. 2016;35:1-23. DOI: 10.1016/j.healun.2015.10.023
- Mehra MR, Uriel N, Naka Y, et al. A Fully Magnetically Levitated Left Ventricular Assist Device – Final Report. N Engl J Med. 2019;380:1618-1627. DOI: 10.1056/NEJMoa1900486
- Mentz RJ, Anstrom KJ, Eisenstein EL, et al. Effect of Torsemide vs Furosemide After Discharge on All-Cause Mortality in Patients Hospitalized With Heart Failure: The TRANSFORM-HF Randomized Clinical Trial. JAMA. 2023;329(3):214-223. DOI: 10.1001/jama.2022.23924
- Mentz RJ, Garg J, Rockhold FW, et al. Ferric Carboxymaltose in Heart Failure with Iron Deficiency. N Engl J Med. 2023;389(11):975-986. DOI: 10.1056/NEJMoa2304968
- MERIT-HF Study Group. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet. 1999;353:2001-2007. DOI: 10.1016/S0140-6736(99)04440-2
- Moss AJ, Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346:877-883. DOI: 10.1056/NEJMoa013474
- Mueller C, McDonald K, de Boer RA, et al. Heart Failure Association of the European Society of Cardiology practical guidance on the use of natriuretic peptide concentrations. Eur J Heart Fail. 2019;21:715-731. DOI: 10.1002/ejhf.1493
- Mullens W, Dauw J, Martens P, et al. Acetazolamide in Acute Decompensated Heart Failure with Volume Overload. N Engl J Med. 2022;387(13):1185-1195. DOI: 10.1056/NEJMoa2203094
- Murphy SL, et al. Genetics of heart failure. Cardiol Clin. 2020;38(1):1-13. DOI: 10.1016/j.ccl.2019.09.001
- Packer M. The neurohormonal hypothesis: a theory to explain the mechanism of disease progression in heart failure. J Am Coll Cardiol. 1992;20:248-254. DOI: 10.1016/0735-1097(92)90167-L
- Packer M, Anker SD, Butler J, et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N Engl J Med. 2020;383:1413-1424. DOI: 10.1056/NEJMoa2022190
- Packer M, Coats AJS, Fowler MB, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001;344:1651-1658. DOI: 10.1056/NEJM200105313442201
- Perera D, Clayton T, O’Kane PD, et al. Percutaneous Revascularization for Ischemic Left Ventricular Dysfunction. N Engl J Med. 2022;387(15):1351-1360. DOI: 10.1056/NEJMoa2206606
- Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med. 1999;341:709-717. DOI: 10.1056/NEJM199909023411001
- Pocock SJ, Ariti CA, McMurray JJV, et al. Predicting survival in heart failure: a risk score based on 39,372 patients from 30 studies. Eur Heart J. 2013;34:1404-1413. DOI: 10.1093/eurheartj/ehs337
- Ponikowski P, Kirwan BA, Anker SD, et al. Ferric carboxymaltose for iron deficiency at discharge after acute heart failure: a multicentre, double-blind, randomised, controlled trial (AFFIRM-AHF). Lancet. 2020;396:1895-1904. DOI: 10.1016/S0140-6736(20)32339-4
- Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2016;37:2129-2200. DOI: 10.1093/eurheartj/ehw128
- Savarese G, Becher PM, Lund LH, et al. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc Res. 2022;118(17):3272-3287. DOI: 10.1093/cvr/cvac013
- Shen L, Jhund PS, Petrie MC, et al. Rapid Guideline-Directed Medical Therapy Titration in Heart Failure. JACC Heart Fail. 2022;10:1-12. DOI: 10.1016/j.jchf.2021.09.006
- SOLVD Investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med. 1991;325:293-302. DOI: 10.1056/NEJM199108013250501
- Stone GW, Lindenfeld J, Abraham WT, et al. Transcatheter Mitral-Valve Repair in Patients with Heart Failure. N Engl J Med. 2018;379:2307-2318. DOI: 10.1056/NEJMoa1806640
- Swedberg K, Komajda M, Böhm M, et al. Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study. Lancet. 2010;376:875-885. DOI: 10.1016/S0140-6736(10)61198-1
- Taylor AL, Ziesche S, Yancy C, et al. Combination of isosorbide dinitrate and hydralazine in blacks with heart failure. N Engl J Med. 2004;351:2049-2057. DOI: 10.1056/NEJMoa042934
- Ter Maaten JM, Beldhuis IE, van der Meer P, et al. Natriuresis-guided diuretic therapy in acute heart failure: a pragmatic randomized trial. Nat Med. 2023;29(10):2625-2632. DOI: 10.1038/s41591-023-02532-z
- Vaduganathan M, Claggett BL, Jhund PS, et al. Estimating lifetime benefits of comprehensive disease-modifying pharmacological therapies in patients with heart failure with reduced ejection fraction. Lancet. 2020;396:121-128. DOI: 10.1016/S0140-6736(20)30748-0
- Velazquez EJ, Lee KL, Jones RH, et al. Coronary-artery bypass surgery in patients with ischemic cardiomyopathy. N Engl J Med. 2016;374:1511-1520. DOI: 10.1056/NEJMoa1602001
- Velazquez EJ, Morrow DA, DeVore AD, et al. Angiotensin-Neprilysin Inhibition in Acute Decompensated Heart Failure (PIONEER-HF). N Engl J Med. 2018;380:539-548. DOI: 10.1056/NEJMoa1812851
- Virani SS, Alonso A, Aparicio HJ, et al. Heart Disease and Stroke Statistics-2021 Update: A Report From the American Heart Association. Circulation. 2021;143:e254-e743. DOI: 10.1161/CIR.0000000000000950
- Wang CS, FitzGerald JM, Schulzer M, et al. Does this dyspneic patient in the emergency department have congestive heart failure? JAMA. 2005;294:1944-1956. DOI: 10.1001/jama.294.15.1944
- Zannad F, McMurray JJV, Krum H, et al. Eplerenone in Patients with Systolic Heart Failure and Mild Symptoms. N Engl J Med. 2011;364:11-21. DOI: 10.1056/NEJMoa1009492

