Cardiovascular medicine is currently witnessing a paradigm shift. For decades, the management of type 2 diabetes mellitus (T2DM) and obesity was siloed, with clinicians focusing primarily on hemoglobin A1c (HbA1c) reduction, blood pressure, lipids and smoking cessation. Weight loss was considered extremely difficult across patient categories. However, the emergence of incretin-based therapies, starting with glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and evolving into multi-receptor agonists, has unleashed a new paradigm in cardiometabolic medicine. These therapies are revolutionizing public health strategies, transcending their original roles as glucose-lowering agents to become essential tools for cardiovascular risk reduction, offering profound benefits in major adverse cardiovascular events (MACE), heart failure with preserved ejection fraction (HFpEF), chronic kidney disease (CKD), and peripheral artery disease (PAD).
Figure 1 (below) shows weight loss effects of novel multi-agonists in recent clinical trials (Rawshani et al).
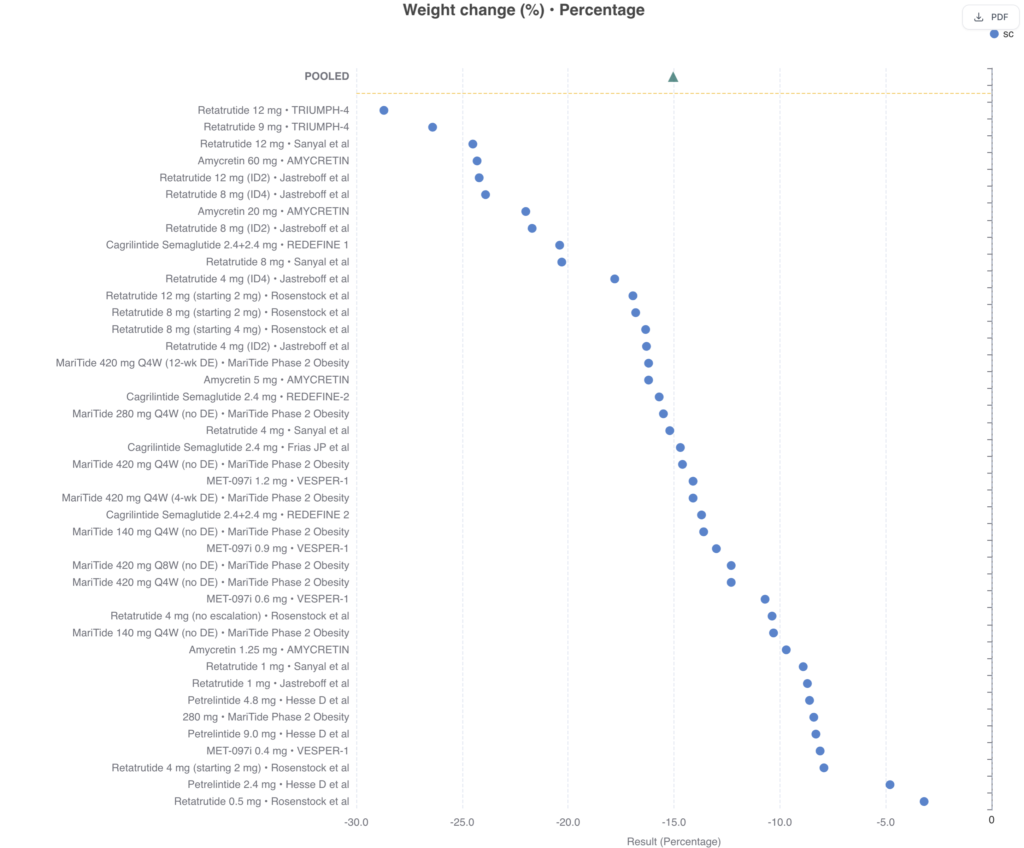
Recommended reading: MetDis Database of Obesity Trials.
Obesity and Cardiovascular Outcomes
The global burden of obesity is increasingly defined by its cardiovascular consequences. In 2015, high body mass index (BMI) was linked to approximately 4.0 million deaths worldwide, with more than two-thirds—roughly 67%—of these fatalities resulting from cardiovascular disease (CVD).1 While BMI remains the most widely utilized metric for assessing weight, its clinical utility is frequently limited by its inability to distinguish between muscle mass and various fat depots, or to identify the precise location of excess adiposity.2
Clinical guidelines now emphasize that a BMI within the conventional “normal” range (up to 25 kg/m²) does not necessarily equate to optimal cardiovascular health.2 Evidence suggests that a lower BMI, potentially around 21 kg/m², may represent a more favorable profile, as the upper limit of the “healthy” range can still harbor significant metabolic risk.3 For patients with a BMI below 35 kg/m², direct assessments such as waist circumference, waist-to-hip ratio, or dual X-ray absorptiometry are recommended to more accurately diagnose clinical obesity and assess cardiovascular risk.4 Furthermore, ectopic fat accumulation, such as intermuscular adipose tissue (IMAT), is independently associated with coronary microvascular dysfunction and major adverse cardiovascular events (MACE), whereas BMI alone often fails to capture these specific pathological drivers.5
Physiological Mechanisms of Cardioprotection: Direct vs. Weight-Mediated
The profound cardiovascular benefits observed with incretin-based therapies cannot be explained by glucose lowering alone. Meta-analyses have shown that the risk reduction in MACE is largely independent of baseline HbA1c or the magnitude of HbA1c reduction.7 Instead, these agents exert multifaceted effects through direct activation of receptors in target tissues.15
Recent evidence clarifies that while the benefits in conditions like osteoarthritis and obstructive sleep apnea primarily reflect the extent of weight loss, the reductions in hard cardiovascular outcomes (MI, stroke) and metabolic liver disease involve significant weight-independent pathways.15
Metabolic Reprogramming and Vascular Health
GLP-1 RAs facilitate “metabolic reprogramming,” shifting myocardial energy metabolism toward more efficient pathways. By modulating fatty acid uptake and mitochondrial function, these agents increase cardiac efficiency. Furthermore, they directly target the vasculature:
- Endothelial Function: Activation of GLP-1 receptors in the endothelium increases nitric oxide bioavailability.10
- Anti-Inflammatory Signaling: These agents inhibit pro-inflammatory pathways (NF-κB and MAPK) in vascular smooth muscle cells, reducing cytokines like IL-6 and TNF-α.11
- Plaque Stability: By reducing macrophage infiltration and inhibiting foam cell formation, they stabilize the fibrous cap of atherosclerotic plaques, preventing rupture.15
The Role of GIP and Glucagon Agonism
Dual agonists like tirzepatide incorporate GIP receptor (GIPR) activation, which increases lipoprotein lipase (LPL) activity in adipocytes, promoting healthy lipid storage and reducing ectopic fat accumulation.12 Newer “triple-action” agents (e.g., retatrutide) incorporate glucagon receptor (GCGR) activity, which increases energy expenditure through hepatic thermogenesis and promotes fat oxidation.15
Cardiovascular and Mortality Outcomes
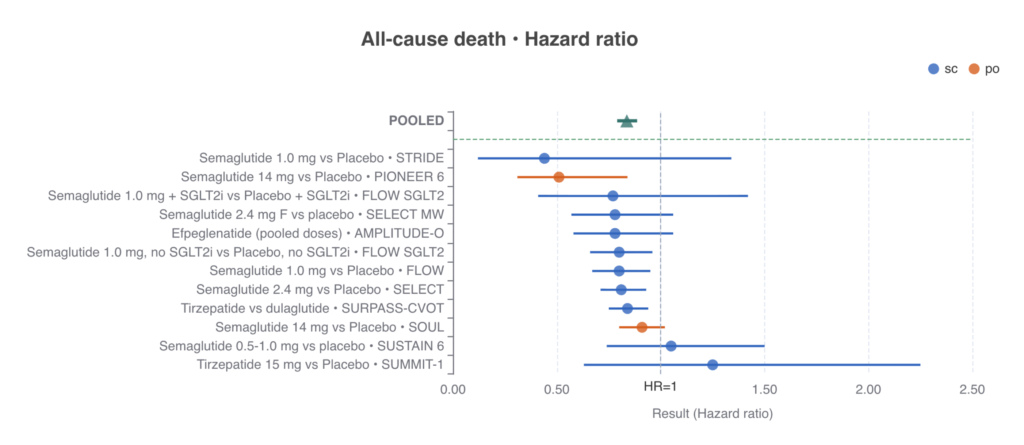
A pooled analysis demonstrates a significant 19% reduction in the risk of all-cause death (HR 0.81; 95% CI 0.72–0.92).
- Route Consistency: This survival advantage is cohesive across both subcutaneous and oral preparations, including oral semaglutide (PIONEER-6, SOUL) and various injectable agents (STRIDE, FLOW, SELECT).
- Broad Efficacy: The benefit extends from GLP-1 mono-agonists to GIP/GLP-1 dual agonists like tirzepatide, which reduced all-cause mortality by 16% compared to dulaglutide in the SURPASS-CVOT trial.15
Atherothrombotic Protection: MACE
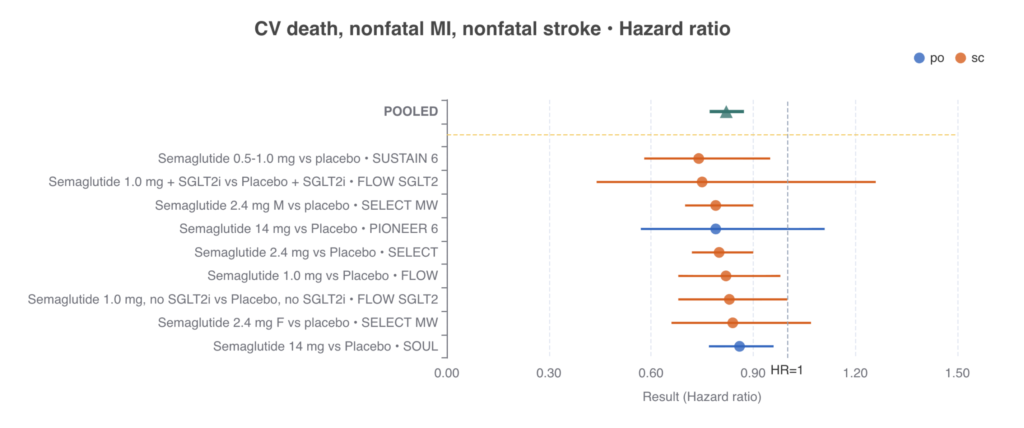
Pooled analyses of 3-point MACE (CV death, nonfatal MI, and nonfatal stroke) reveal a robust 21% reduction in major adverse cardiovascular events (HR 0.79; 95% CI 0.72–0.87). This cardioprotective effect is notably independent of dosing intensity or mode of administration. Furthermore, the benefit remains uniform even in the presence of other foundational therapies, such as SGLT2 inhibitors.
Anti-Inflammatory Effects and Biomarkers
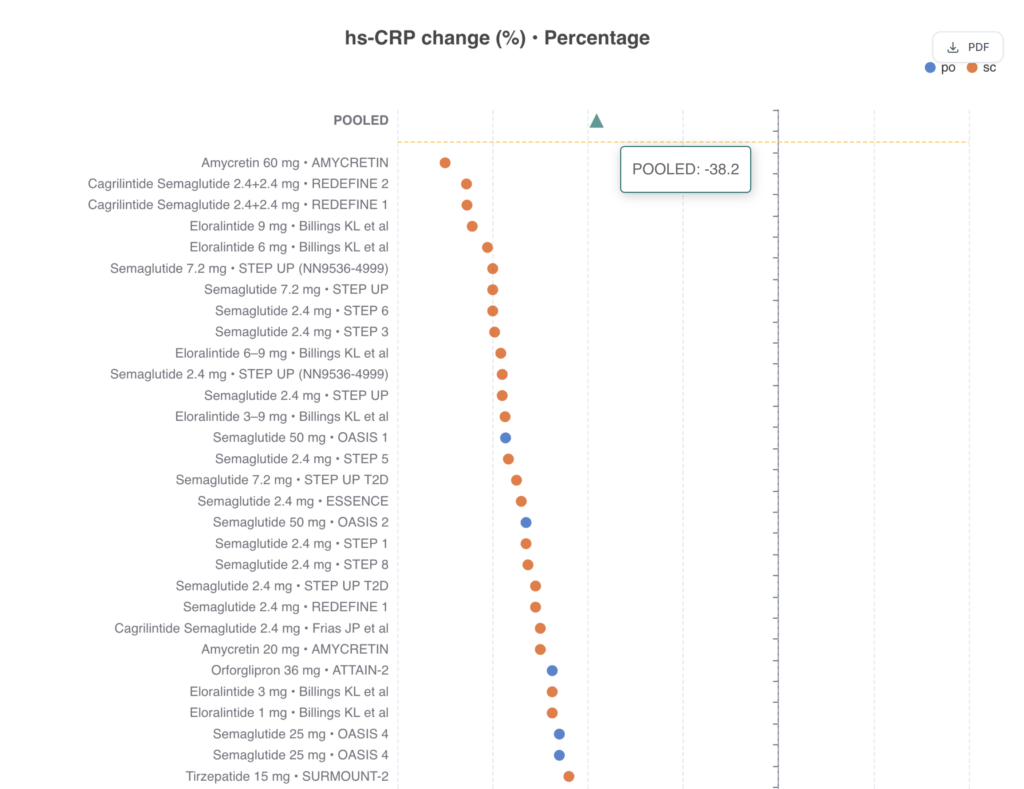
A hallmark of incretin therapy is the robust attenuation of systemic inflammation. Pooled estimates across clinical trial programs demonstrate a striking 43.1% reduction in high-sensitivity C-reactive protein (hsCRP) levels.
- Combination Efficacy: The highest reductions are reported with combination therapies like CagriSema (cagrilintide/semaglutide) and triple agonists like retatrutide.15
- Weight Independence: Evidence suggests that the magnitude of hsCRP reduction is only partially mediated by weight loss, pointing to a direct immunomodulatory effect on immune cells.15
PAD, OSA, and MASH
The utility of incretins has expanded rapidly beyond the traditional T2DM/obesity:
- Peripheral Artery Disease (PAD): The STRIDE trial demonstrated that semaglutide improves walking capacity in patients with symptomatic PAD and T2DM.15
- Obstructive Sleep Apnea (OSA): Tirzepatide significantly improved the apnea-hypopnea index in patients with OSA and obesity, primarily through profound weight reduction.15
- Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD/MASH): Trial data for semaglutide and survodutide show biopsy-proven resolution of MASH and potential improvements in liver fibrosis.15
The SELECT Trial: Rapid Cardioprotection
The SELECT trial (17,604 non-diabetic patients with obesity and CVD) showed a 20% reduction in MACE.14 Crucially, significant benefits were evident within the first 6 months, before maximum weight loss occurred.
- The Paradox: Analysis revealed an HR of 0.58 at 20 weeks. Weight reduction accounted for virtually none of this early benefit, whereas reduction in waist circumference (visceral fat) explained only 33% of the overall effect. The remaining 67% is attributed to direct vascular and anti-inflammatory pleiotropic mechanisms.15
The Next Frontier: Multi-Agonists and Small Molecules
| Drug | Mechanism | Status/Result |
| Tirzepatide | GLP-1R / GIPR Agonist | Superior weight loss and glycemic control vs. semaglutide 15 |
| CagriSema | Amylin / GLP-1R Agonist | 17.3% weight loss in REDEFINE-1; consistent safety 15 |
| Maritide | GIPR Antagonist / GLP-1RA | Once-monthly dosing; 17.3% weight loss in phase 2 15 |
| Retatrutide | GIPR / GCGR / GLP-1RA | Up to 24.2% weight loss; potential for energy expenditure increase 15 |
| Orforglipron | Oral Small-Molecule GLP-1RA | -1.6% HbA1c and 12.4% weight loss; needle-free alternative 15 |
| Amycretin | Oral GLP-1 / Amylin Agonist | Up to 13.1% weight loss over only 12 weeks 15 |
Clinical Challenges and Precision Medicine
Muscle Health and Sarcopenic Obesity
Extensive weight loss (25-35%) with newer agents is often accompanied by 20-40% reductions in lean mass. Investigational “muscle-sparing” agents, such as bimagrumab (activin/myostatin inhibitor), are being studied in combination with GLP-1 RAs to ensure that weight loss is “healthy” and functional.15
Heterogeneity of Response
Considerable variability exists in weight loss response, with some patients losing $>20\%$ and others gaining weight. Currently, genetic variants in $GLP1R$ or $ARRB1$ (encoding $\beta$-arrestin) explain only marginal differences in response, and no biomarkers yet accurately predict who will be a “super-responder”.15
Safety and Long-term Management
While generally safe, clinicians must monitor for:
- Gastrointestinal (GI) Events: Nausea and diarrhea are common but typically resolve with slow titration. Multi-agonists like tirzepatide may utilize GIP’s “anti-aversive” actions to improve GI tolerability relative to the degree of weight loss.15
- Kidney Health: Sustained GI side effects can lead to dehydration and acute kidney injury (AKI), warranting close monitoring in high-risk patients.15
- Diabetic Retinopathy: Rapid glucose lowering can cause transient worsening. The randomized FOCUS trial is currently investigating semaglutide’s long-term ocular effects.15
- Pregnancy: While stopping GLP-1 RAs before conception is standard, registry data of inadvertent first-trimester exposure have not shown evidence of teratogenicity or major birth defects.15
Conclusion
The integration of GLP-1 RAs and multi-agonists represents the most significant advancement in cardiometabolic risk reduction of the 21st century. We have moved from a model of reactive management to a proactive strategy of addressing the common metabolic drivers of heart failure, stroke, and chronic kidney disease. As we move toward more powerful combinations and oral non-peptides, the goal for the cardiovascular team is now to ensure equitable access to these life-saving tools and to transition from treating “obesity” to treating “cardiometabolic disease.”
References
- Health Effects of Overweight and Obesity in 195 Countries over 25 …, accessed on January 12, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC5477817/
- American College of Cardiology Issues Guidance on Weight …, accessed on January 12, 2026, https://www.acc.org/About-ACC/Press-Releases/2025/06/20/14/11/American-College-of-Cardiology-Issues-Guidance-on-Weight-Management-Drugs
- 2025 Concise Clinical Guidance: An ACC Expert Consensus Statement on Medical Weight Management for Optimization of Cardiovascular Health – Portail Vasculaire, accessed on January 12, 2026, https://www.portailvasculaire.fr/sites/default/files/docs/gilbert-et-al-2025-concise-clinical-guidance-an-acc-expert-consensus-statement-on-medical-weight-management-for.pdf
- Study Details | NCT06383390 | The Effect of Retatrutide Once Weekly on Cardiovascular Outcomes and Kidney Outcomes in Adults Living With Obesity (TRIUMPH-Outcomes) | ClinicalTrials.gov, accessed on January 12, 2026, https://www.clinicaltrials.gov/study/NCT06383390
- The Cardiovascular Effect of Tirzepatide: A Glucagon-Like Peptide-1 and Glucose-Dependent Insulinotropic Polypeptide Dual Agonist – NIH, accessed on January 12, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC10548186/
- Semaglutide and Cardiovascular Outcomes – NEJM | Request PDF – ResearchGate, accessed on January 12, 2026, https://www.researchgate.net/publication/378366495_Semaglutide_and_Cardiovascular_Outcomes_-_NEJM
- GLP-1 receptor agonists and cardiorenal outcomes in type 2 … – NIH, accessed on January 12, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC8442438/
- Semaglutide and Cardiovascular Outcomes by Baseline HbA1c and Change in HbA1c in People With Overweight or Obesity but Without Diabetes in SELECT, accessed on January 12, 2026, https://diabetesjournals.org/care/article/47/8/1360/156810/Semaglutide-and-Cardiovascular-Outcomes-by
- Semaglutide in obesity-related heart failure with preserved ejection fraction and type 2 diabetes across baseline HbA1c levels (STEP-HFpEF DM): a prespecified analysis of heart failure and metabolic outcomes from a randomised, placebo-controlled trial – PubMed, accessed on January 12, 2026, https://pubmed.ncbi.nlm.nih.gov/39848268/
- GLP-1 Agonists in Cardiovascular Diseases: Mechanisms, Clinical …, accessed on January 12, 2026, https://www.mdpi.com/2077-0383/14/19/6758
- Glucagon-like peptide-1 receptor agonists: a review from a cardiovascular perspective, accessed on January 12, 2026, https://www.frontiersin.org/journals/cardiovascular-medicine/articles/10.3389/fcvm.2025.1535134/full
- Mechanisms of action and therapeutic applications of GLP-1 and dual GIP/GLP-1 receptor agonists – Frontiers, accessed on January 12, 2026, https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2024.1431292/full
- Reduction of prevalence of patients meeting the criteria for metabolic syndrome with tirzepatide: a post hoc analysis from the SURPASS Clinical Trial Program – PubMed Central, accessed on January 12, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC10859014/
- Semaglutide Effects on Cardiovascular Outcomes in People With …, accessed on January 12, 2026, https://www.acc.org/Latest-in-Cardiology/Clinical-Trials/2023/11/09/15/04/select
- The expanding landscape of GLP-1 medicines. D. Drucker. Nature Medicine.

